Answers
Answer:
Mr.clark is prescribed 500 mg of acetaminophen TID and 30 ml of prednisone BID. Mr.clark takes his first dose of both medications at 0800 hours when would you administer the second dose of acetaminophen?
Mr. Clark is prescribed 500 mg of acetaminophen TID and 30 ml of prednisone BID. Mr. Clark takes his first dose of both medications at 0800 hours. [ You would administer the second dose of acetaminophen at 1400 hours. ]
Related Questions
HELP ASAP PLEASE. A legend is a traditional story that explains something in real life. Sometimes they are accepted as true, but sometimes they are fantastical and just plain fun. There are many legends that surround the North Star explaining its creation as well as why it is stationary.
Take some time to research some of the legends of the North Star. After you have had a chance to read several of them, create your own fantastical story of the North Star. It can be how it came to be or explain why it is stationary.
Answers
A legend of the North star goes as follows:
Once upon a time, when God was creating the universe, the curious star Tam once he learned that God was humans on earth, he began visiting humans. He became fond of one human. But each time he had to return to the sky, the human could not see him again. Therefore, he decided to remain stationary so that the human can always locate him.What is the North Star?The North star is a star that sits directly above the North pole of the earth. It is also known as Polaris.
The North star is especially significant in that the North star is an ever-present star in the skies. It neither rises nor sets unlike other stars in the skies.
Because of this special feature of the North star, it serves as a guiding beacon in the skies for travelers. Also, several legends have been told about the North star.
Learn more about the North star at: https://brainly.com/question/1130377
#SPJ1
What molecule is this?

Answers
Answer:
O2C
Explanation:
Answer:
Carbon Monoxide (CO) is a colorless, odorless, tasteless, toxic gas that has the molecular formula CO. The molecule consists of a carbon atom that is triply bonded to an oxygen atom. ... The linear molecule consists of a carbon atom that is doubly bonded to two oxygen atoms, O=C=O
Explanation:
Separating techniques - Chromatography
Someone has written rude words in graffiti ink all over the back bench in the Science
classroom where three students usually sit. Miss has said the whole class will forfeit
their lunchtime unless you can find out who the culprit is.
Your task is to plan a detailed investigation into the colours in the graffiti ink and the ink
used by the culprits to find out whose ink wrote the graffiti
You should include:
a List of apparatus required
description of the method used
a labelled diagram of your results
any hazards or risks
a conclusion and evaluation
.
Keywords: soluble solvent chromatography chromatogram separate mixtures colours
To get a higher
mark:
You need to have:
.
.
.
.
Described a method of separation of ink using most of the key words.
Drawn and labelled a diagram of results.
Conducted a full risk assessment.
Described how chromatography is useful
Explained your results and drawn a conclusion
Explained how chromatography works and the uses of chromatography
Explained your choice of separation technique, stating other types of separation
techniques
Suggested any possible problems with the method or in using the results.
• Suggested how the investigation could have been improved.
Compared the distance moved and Rf values for the different pigments in different
solvents and used this data to draw conclusions
Evaluated how good the evidence was in supporting the conclusion
Answers
The method that can be used to separate the mixture is chromatography.
What is chromatography?
"Chromatography" is obtained form a Greek word which literarily means, color writing. It is a method of separation which is common in separating a mixture of pigments.
To obtain the colors used, two solvents are mixed and the sample ink is dissolved in the solvents then spotted on a thin layer and put into a TLC chamber then the chromatogram is allowed to develop.
The various components of the pigment will appear on the chromatogram and can be identified using spectrophotometry. The Rf values of each component can also be used to identify it.'
Learn more about chromatography: https://brainly.com/question/26491567
What is Markanikov rule?
Answers
Answer:
Regla de Markovnikov En fisicoquímica orgánica, la regla de Markovnikov es una observación respecto a la reacción de adición electrófila.Regla de Markovnikov En fisicoquímica orgánica, la regla de Markovnikov es una observación respecto a la reacción de adición electrófila.
Explanation:
can be used to help an individual with an eating disorder learn better ways to cope with stress an
replace unhealthy behaviors with those that do not cause harm.
A. Preventative care
B. Hospitalization
C. Medication
D. Psychotherapy
Answers
Answer: Psychotherapy
Explanation:
Edge
Answer:
D. Psychotherapy
Explanation:
Did the test
can iodine dissolve in acetone
Answers
Answer:
no it wont dissolves
Explanation:
iodine will only dissolve in solvents that lack of polarity. acetone is a strongly polar solvent. therefore iodine would not disslove in acetone.
hope this helps!
Why would you not want to use a salt bridge saturated with potassium chloride solution in an electro-chemical cell made from a Ag/Ag+ cathode and a Cu/Cu²+ anode?
Answers
We can not use a salt bridge saturated with potassium chloride solution in an electro-chemical cell made from a Ag/Ag+ cathode and a Cu/Cu²+ anode because it will precipitate.
An electrochemical cell that uses a weak electrolyte and a salt bridge to connect oxidation as well as reduction half cells. A junction that joins the anodic with cathodic compartments of a cell and electrolytic solution is referred to as a salt bridge.
Because both chloride and potassium ions have very similar diffusion coefficients and minimise junction potential, the inactive minerals potassium chloride (KCl) frequently used. We can not use a salt bridge saturated with potassium chloride solution in an electro-chemical cell made from a Ag/Ag+ cathode and a Cu/Cu²+ anode because it will precipitate.
To know more about salt bridge, here:
https://brainly.com/question/2861410
#SPJ1
inorganic compound created by the removal or replacement of one, two, or all three hydrogen atoms in phosphoric acid; used in fertilizers and detergents and is a major cause of water pollution
Answers
The inorganic compound that is created by the removal or replacement of one, two, or all three hydrogen atoms in phosphoric acid and is commonly used in fertilizers and detergents, as well as a major cause of water pollution, is called "phosphate."
What do you mean by inorganic compound?
An inorganic compound is a chemical compound that does not contain carbon-hydrogen (C-H) bonds, which are the defining characteristic of organic compounds. Inorganic compounds can be composed of a variety of elements, including metals, non-metals, and metalloids.
The inorganic compound that is created by the removal or replacement of one, two, or all three hydrogen atoms in phosphoric acid and is commonly used in fertilizers and detergents, as well as a major cause of water pollution, is called "phosphate."
Phosphate compounds are essential nutrients for plant growth and are often added to fertilizers to improve soil fertility. However, when excess phosphate is added to water bodies, it can lead to eutrophication, a process where an excessive growth of algae and other aquatic plants occur, ultimately leading to oxygen depletion and harm to aquatic life.
Phosphates are also commonly used in detergents to aid in cleaning, but they can have similar negative impacts on water quality if they are not properly treated before being discharged into waterways. Therefore, regulations are in place to limit the amount of phosphate that can be added to detergents and other products in many countries to mitigate water pollution.
Learn more about inorganic compound click here:
https://brainly.com/question/26221159
#SPJ1
A 12.2 mL sample of liquid was found to have a mass of 10.4 g. Calculate the density of this liquid ( in g/mL).
Answers
Answer:
d=m/
Explanation:
d is density, m is mass, v is volume
Given: m =10.4g, v=12.2mL
substituting in equation,
d=10.4/ 12.2
d=0.8524g/mL
To learn more about density:
The density of the liquid is 0.852 g/mL.
To calculate the density of the liquid, we need to use the formula:
Density = Mass / Volume
Given that the mass of the liquid is 10.4 g and the volume is 12.2 mL, we can substitute these values into the formula:
Density = 10.4 g / 12.2 mL
Simplifying this expression, we find:
Density = 0.852 g/mL
Density is a physical property of a substance and is defined as the amount of mass per unit volume. In this case, the density tells us that for every milliliter of the liquid, there is 0.852 grams of mass. The units of grams per milliliter (g/mL) indicate that the density is a ratio of mass to volume.It is important to note that the density of a substance can vary with temperature, so this value is only valid under the conditions at which the measurement was made. Additionally, the density can provide valuable information about the identity of a substance, as different substances have different densities.
for such more questions on density
https://brainly.com/question/26364788
#SPJ8
What is the formula for the carboxylic acid functional group?
OA. -CH3
OB. -COH
0 с. -соон
OD. -CO-
Answers
Answer: C
Explanation:
This is a fact.
How much heat is required to melt 38.6 g of ice (0 degrees Celsius)? The heat of fusion for water is 6.02 kJ/mol.
Express your answer in kilojoules to three significant figures
Answers
Given the heat of fusion of water is 6.02 kJ/mol, it takes 12.9 kJ of heat to melt 38.6 g of ice at 0 °C.
What is fusion?Fusion, commonly known as melting, is a physical process in which matter passes from the solid to the liquid state.
We have ice with a mass of 38.6 g at 0 °C. First, we will convert mass to moles using the molar mass of water.
38.6 g × (1 mol/18.02 g) = 2.14 mol
The heat of fusion (ΔH°fus) for water is 6.02 kJ/mol. The heat (Q) required to melt 2.14 moles of ice is:
Q = 6.02 kJ/mol × 2.14 mol = 12.9 kJ
Given the heat of fusion of water is 6.02 kJ/mol, it takes 12.9 kJ of heat to melt 38.6 g of ice at 0 °C.
Learn more about fusion here: https://brainly.com/question/40140
#SPJ1
Consider the reaction
2NO(g) + O2(g) = 2NO2(g)
Suppose that at a particular moment during the reaction nitric oxide
(NO) is reacting at the rate of 0.066 M/s. (a) At what rate is NO2
being formed? (b) At what rate is molecular oxygen reacting?
Answers
Answer:
(a) Rate of formation of NO2 is also 0.066M/s
(b) Rate of reaction of O2 gas is 0.033M/s
Explanation:
(a) in one second, according to the equation,
2 moles of NO combines with 2moles of NO2.
Therefore 0.066M NO will still consume 0.066mole NO2.
(b) According to the equation,
2 moles NO consumes 1 mole O2, 0.0666M will consume 0.0333 mole O2
Give the electron configurations for the following ions: Al3+, Ba2+, Br- and O2-
Answers
Explanation:
Al3+ has 10 electrons. The electronic configuration is;
1s² 2s² 2p⁶
Ba2+ has 54 electrons. The electronic configuration is;
1s² 2s² 2p6 3s² 3p⁶ 3d10 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶
Br- has 37 electrons. The electronic configuration is;
1s² 2s² 2p6 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶
O2- has 10 electrons. The electronic configuration is;
1s² 2s² 2p⁶
NEED HELP ASAP!!!!!!!!! The process labeled #6 in this picture is known as
A. endocytosis
B. exocytosis
C. passive transport
D. diffusion

Answers
Answer:
B - Exocytosis
Explanation:
Exocytosis is the process by which a large amount of molecules are released; thus it is a form of bulk transport. In exocytosis, membrane-bound secretory vesicles are carried to the cell membrane, and their contents are secreted into the extracellular environment.
Complete the table. Then explain how wavelength and frequency are mathematically related.
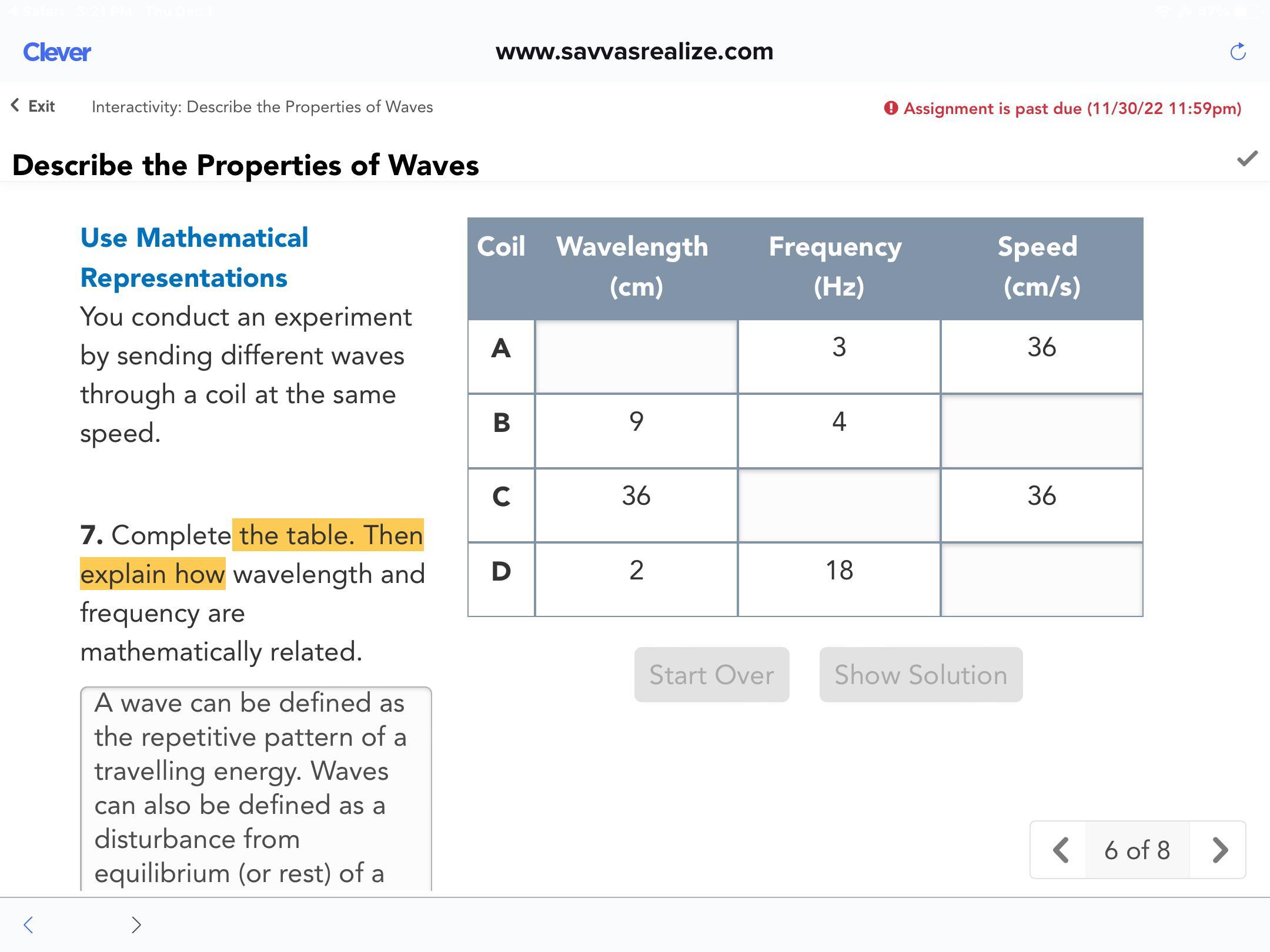
Answers
The wavelength is same.
What is wavelength?
Wavelength can be defined as the distance between two successive by throughs of a wave.
The question wants you to determine the energy that is the incoming photon by the must have in order to the accept the electron that melted it tob the jump to 2 to 1.
A good starting point here will be to calculate the energy of to the photon emitted when the electron falls from 1 to 2
by applying the Rydberg equation.
1/π = R
λ
si the wavelength of the emittted photon R
is the Rydberg constant, equal to 1.907.
This means that you haveλ=
4.10
So, you know that when an electron falls from the
to
a photon of wavelength by the
410 nm
is emitted. This implies by that in order to the for the electron by jump to the 2 to 1 .
it must absorb a photon of the same wavelength.
To know more about wavelentgh click-
https://brainly.com/question/10750459
#SPJ1
A solution contains 6 mg isopropanol dissolved in 2 kg H2O. The isopropanol concentration is?
a. 3% isopropanol.
b.3 M isopropanol.
c.3 m isopropanol.
d. 3 ppm isopropanol.
Answers
Answer:
Option D. 3 ppm isopropanol.
Explanation:
From the question given above, the following data were obtained:
Mass of isopropanol = 6 mg
Mass of water = 2 Kg
Isopropanol concentration =?
Concentration = mass isopropanol / mass of water
Isopropanol concentration = 6 mg / 2 kg
Isopropanol concentration = 3 mg/Kg
Next, we shall convert 3 mg/Kg to ppm. This can be obtained as follow:
Recall:
1 mg/Kg = 1 ppm
Therefore,
3 mg/Kg = 3 ppm
Thus, the isopropanol concentration is 3 ppm.
What state of matter can be compressed?
A. gases
B. solids, liquids, and gases
C. liquids
D. solids
Answers
Answer:
Liquids, They are the only one that can change shape to fit the container
Answer:
option A is correct.
Explanation:
The atoms and molecules in gases are much more spread out than in solids or liquids. They vibrate and move freely at high speeds. A gas will fill any container, but if the container is not sealed, the gas will escape. Gas can be compressed much more easily than a liquid or solid.
The volume of a sample of gas (2.49 g) was 752 mL at 1.98 atm and 62°C. The gas is most likely __________.
A. SO2
B. SO3
C. NH3
D. NO2
E. Ne
Answers
The gas that has a volume of 752 mL at 1.98 atm and 62°C is most likely NO₂ (option D).
How to calculate volume?The volume of a sample of gas can be calculated using the following formula:
PV = nRT
Where;
P = pressureV = volume n = number of molesR = gas law constantT = temperatureAccording to this question, the volume of a sample of gas (2.49 g) was 752 mL at 1.98 atm and 62°C. The number of moles is as follows:
1.98 × 0.752 = n × 0.0821 × 335
1.489 = 27.5n
n = 0.054mol
molar mass of the gas = 2.49g ÷ 0.054mol = 45.99g/mol
The gaseous substance with the molar mass of 45.99g/mol is NO₂.
Learn more about moles at: https://brainly.com/question/27058396
#SPJ1
PLEASE HELP QUICKLY!!!
HI gas is removed from the system
at equilibrium below. How does the
system adjust to reestablish
equilibrium?
51.8 kJ + H₂(g) + 1₂(g) = 2HI(g)
A. The reaction shifts to the right (products) and the concentrations
of I, and H₂ decrease.
B. The reaction shifts to the left (reactants) and the concentrations
of H₂ and I increase.
C. The reaction shifts to the right (products) and the concentrations
of I, and H₂ increase.
D. The reaction shifts to the left (reactants) and the concentration of
HI increases.

Answers
Answer:
A. The reaction shifts to the right (products) and the concentrations of I and H₂ decrease.
Explanation:
If gas is removed from the system at equilibrium, the system will try to compensate for the loss by shifting the reaction in a direction that produces more gas molecules. This is known as Le Chatelier's principle, which states that a system at equilibrium will respond to a disturbance by shifting in a way that minimizes the effect of the disturbance.
In this case, since gas is being removed from the system, the reaction will shift to the side that produces more gas molecules. Looking at the balanced equation, we can see that 2HI(g) has a greater number of gas molecules compared to H₂(g) and I₂(g). Therefore, the system will shift to the right (products) to produce more HI(g) and reestablish equilibrium.
Sodium is a reactive, alkali metal. A neutral sodium atom consists of 11 protons, 11 electrons and 12 neutrons. It is a soft metal and metallic silver in color. Sodium plays an important role in the functioning of the human nervous system.
Which of the following images represents the atomic structure of a neutral sodium atom?
W.
X.
Y.
Z.
A.
W
B.
X
C.
Z
D.
Y
Answers
Sodium is a reactive, alkali metal. A neutral sodium atom consists of 11 protons, 11 electrons, and 12 neutrons. the atomic structure of a neutral sodium atom is represented by Y. Option D
What is atomic structure?Generally, Atomic structure is the structure of an atom, which consists of a nucleus (the center) and protons (which are positively charged), and neutrons (which are neutral).
This structure is referred to as the structure of an atom. Electrons are negatively charged particles that orbit the nucleus, which is located at the center of the atom.
In conclusion, Alkali sodium is reactive. Sodium has 11 protons, 11 electrons, and 12 neutrons. Neutral sodium's atomic structure is Y.
Read more about atomic structure
https://brainly.com/question/13089956
#SPJ1
Which of the following could not act as a medium for a mechanical wave?
a) air
b) empty space
c) liquid water
d) a solid rope
Answers
Answer:
b) empty space
Explanation:
A mechanical cannot travel through empty space. So option (b) is correct.
A mechanical wave is a wave which needs a material medium for its propagation. For example sound, water waves etc . The medium required by the wave can be a solid, liquid or a gas. Empty space doesn't have any medium, so a mechanical wave cannot travel through empty space.
What is the total number of atoms of magnesium and phosphorus in 3Mg3(PO4)2?
Answers
Thus, 1 molecule of magnesium phosphate will contain 2 atoms of phosphorus.
Therefore, three molecules of magnesium phosphate contains following number of atoms.
Mg = 9
P = 6
O = 24
Hence, we can conclude that there are 6 atoms of phosphorus in three molecules of magnesium phosphate, .
convert 8.42x10^8 mol/(kg*m^2) to mol/(g*cm^2)
Answers
Answer:
gguhg
Explanation:
no te es caso drama me están muy una las y y que las te
1 kg = 1000 g
1 m = 100 cm
Using these equations, 8.42x10^8 mol/(kgm^2) can be converted to 8.42x10^11 mol/(gcm^2).
Explain ocean currents and how density differences between HOT/COLD and SALT/FRESH affect them.
-for science
Answers
Density differences caused by temperature and salinity variations are fundamental drivers of ocean currents of seawater . Warm currents transport heat from the equator to higher latitudes, while cold currents transport cold water from higher latitudes to lower latitudes.
When seawater is heated, it expands and becomes less dense, causing it to rise. Conversely, when seawater cools, it contracts and becomes denser, causing it to sink. These density differences due to temperature variations create vertical movements in the ocean known as thermohaline circulation or convection currents. On the other hand, regions with high freshwater input from rivers or heavy precipitation have lower salinity, resulting in lower density. This lighter water tends to float on the denser seawater beneath it, leading to the formation of surface currents that transport water from areas of low salinity to areas of higher salinity. These ocean currents play a vital role in shaping global climate patterns and maintaining the balance of heat and nutrients in the ocean ecosystem.
Learn more about the ocean current here.
https://brainly.com/question/21654036
#SPJ1
Enter an equation showing how this buffer neutralizes added aqueous acid (HI). Express your answer as a chemical equation. Identify all of the phases in your answer.
Answers
Answer:
\(H^++NH_3\rightleftharpoons NH_4^+\)
Explanation:
Hello there!
In this case, since the buffer is not given, we assume it is based off ammonia, it means the ammonia-ammonium buffer, whereas the ammonia is the weak base and the ammonium ion stands for the conjugate acid. In such a way, when adding HI to the solution, the base of the buffer, NH3, reacts with the former to promote the following chemical reaction:
\(H^++NH_3\rightleftharpoons NH_4^+\)
Because the HI is totally ionized in solution so the iodide ion becomes an spectator one.
Best regards!
Answer:
the above answer is wrong
Explanation:
its really not but the mastering chemistry says it is
What is happening to the matter in this system?
Answers
Answer:
good luck maybe A im not smart though
Explanation:
Name the following Type 3 compound
1. PCIE
2. P406
3. SF6
4. SO3
5. SO2
6. N203
7. SiO2
8. O2F2
9. XeF6
Answers
Answer:
32
Explanation:
8.50 of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 160./gmol, is burned completely in excess oxygen, and the mass of the products carefully measured: product mass carbon dioxide 14.03g water 3.83g
Answers
The compound would have a molecular formula of C5H10O2.
What is the empirical formula of the compound?
The empirical formulas is the simplest formula of a compound.
We know that;
Mass of the carbon = 14.03 * 12/44 = 3.82 g
Moles of C = 3.82 g/12 g/mol = 0.32 moles
Mass of water = 3.83 * 2/18 = 0.43 g
Moles of H = 0.43 g/1 g/mol = 0.43 moles
Mass of oxygen = 8.5 - (3.82 + 0.43)
= 4.25 g
Moles of oxygen = 4.25 g/16 g/mol = 0.27 moles
We can now divide through by the lowest ratio;
C - 0.32 H - 0.43 O - 0.27
C - 1 H - 2 O - 1
The empirical formula is CH2O
The molecular formula is;
[12 + 2+ 16]n = 160
n = 160/30
n = 5
The molecular formula is;
C5H10O2
Learn more about molecular formula :https://brainly.com/question/20748250
#SPJ1

A 2.00-mol sample of hydrogen gas is heated at constant pressure from 294 K to 414 K. (a) Calculate the energy transferred to the gas by heat. kJ (b) Calculate the increase in its internal energy. kJ (c) Calculate the work done on the gas. kJ
Answers
Answer:
a) The energy transferred is 6.91 kJ
b) The internal energy is 4.90 kJ
c) The work done on the gas is - 2.01 kJ
Explanation:
Step 1: Data given
Number of moles of hydrogen gas = 2.00 moles
Pressure = constant
Temperature is heated from 294 K to 414 K
Molar heat capacity of hydrogen gas = 28.8 J/mol*K
Step 2: Calculate the energy transferred to the gas by heat.
Q = n* Cp * ΔT
⇒with Q =the energy transferred
⇒with n = the number of moles = 2.00 moles
⇒with Cp = the Molar heat capacity of hydrogen gas = 28.8 J/mol*K
⇒ with ΔT = Temperature 2 - Temperature 1 = 414 - 294 = 120K
Q = 2.00 * 28.8 * 120
Q = 6912 J = 6.91 kJ
Step 3: Calculate the increase in its internal energy.
ΔEint = n*Cv*ΔT
⇒with ΔEint = the increase in its internal energy.
⇒with n = the number of moles = 2.00 moles
⇒with Cv = The constant volume = 20.4 J/mol*K
⇒with ΔT = Temperature 2 - Temperature 1 = 414 - 294 = 120K
ΔEint = 2.00 * 20.4 * 120
ΔEint =4896 J = 4.90 kJ
Step 4: Calculate the work done on the gas.
Work done on the gas = -Q + ΔEint
W = -6.91 kJ + 4.90 kJ
W = -2.01 kJ
What is the basic unit of stucture and function of living things