Answers
Answer:
A. 0.160 M
B. 0.360 M
C. 0.450 M
Explanation:
A. Determination of molarity of Cl- in
0.160 M NaCl.
We shall write the dissociation equation for NaCl. This is illustrated below:
NaCl —> Na+ + Cl-
From the balanced equation above,
1 mole of NaCl produced 1 mole of Cl-
Therefore, 0.160 M NaCl will also produce 0.160 M Cl-
Therefore the molarity of Cl- is 0.160 M
B. Determination of molarity of Cl- in 0.180 M SrCl2
We shall write the dissociation equation for SrCl2. This is illustrated below:
SrCl2 —> Sr^2+ + 2Cl-
From the balanced equation above,
1 mole of SrCl2 produced 2 moles of Cl-
Therefore, 0.180 M SrCl2 will produce = 0.180 x 2 = 0.360 M Cl-
Therefore, the molarity of Cl- is 0.360 M
C. Determination of molarity of Cl- in 0.150 M AlCl3.
We shall write the dissociation equation for AlCl3. This is illustrated below:
AlCl3 —> Al^3+ + 3Cl-
From the balanced equation above,
1 mole of AlCl3 produced 3 moles of Cl-
Therefore, 0.150 M AlCl3 will produce = 0.150 x 3 = 0.450 M Cl-
Therefore, the molarity of Cl- is 0.450 M.
A) The molarity in Cl⁻ 0.160 M NaCl = 0.160 M
B) The molarity in Cl⁻ 0.180 M SrCl₂ = 0.360 M
C) The molarity in Cl⁻ 0.150 M AlCl₃ = 0.450 M
A) Determine the molarity in Cl⁻ 0.160 M NaCl
first step : write out the balanced dissociation reaction equation for NaCl
NaCl ----> Na⁺ + Cl⁻
From the balanced dissociation equation : 1 mole of NaCl = 1 mole of Cl⁻
∴ 0.160 M of NaCl = 0.160 M of Cl⁻
B) Determine The molarity in Cl⁻ 0.180 M SrCl₂
First step : write out the balanced dissociation reaction equation for SrCl₂
SrCl₂ ---> Sr²⁺ + 2Cl⁻
From the equation ; 1 mole of SrCl₂ = 2 moles of Cl⁻
∴ 0.180 M of SrCl₂ = 0.180 * 2 = 0.360 M of Cl⁻
C) Determine the molarity in Cl⁻ 0.150 M AlCl₃
First step : write out the balanced dissociation reaction equation for AlCl₃
AlCl₃ ---> Al³⁺ + 3Cl⁻
From the balanced dissociation equation
0.150 M AlCl₃ will produce = 0.150 * 3 = 0.450 M of Cl⁻
Hence we can conclude that A) The molarity in Cl⁻ 0.160 M NaCl = 0.160 M, The molarity in Cl⁻ 0.180 M SrCl₂ = 0.360 M, The molarity in Cl⁻ 0.150 M AlCl₃ = 0.450 M
Related Questions
What would be the name of this compound?

Answers
Answer:
2,3 Dimethyl hexane
Explanation:
First, start the count from which side is given the CH3 smallest number
first; the longest carbon chain in this compound is 6
and you don't have any double and triple bonds or functional groups so it is Hexane
you start to count from the right side to give the branch molecules the smallest number ..
CH3 = methyl
and you have 2 methyl in this compound ..
and 2 mean you must write ( Di )
you write the name in this way
2,3 Dimethyl hexane
hope this helps you.
stay safe ...
Unit Conversion Help Thank you

Answers
Answer : 1721.72 g/qt are in 18.2 g/cL
Explanation :
As we are given: 18.2 g/cL
Now we have to convert 18.2 g/cL to g/qt.
Conversions used are:
(1) 1 L = 100 cL
(2) 1 L = 1000 mL
(3) 1 qt = 946 qt
The conversion expression will be:
\(\frac{18.2g}{1cL}\times \frac{100cL}{1L}\times \frac{1L}{1000mL}\times \frac{946mL}{1qt}\)
\(=1721.72\text{ g/qt}\)
Therefore, 1721.72 g/qt are in 18.2 g/cL
how many moles of NaOH in 125.0 mL of 0.190M NaOH?
Answers
Answer:
0.0238 moles NaOH
Explanation:
To find the amount of moles, you need to
(1) convert the volume from mL to L (1,000 mL = 1 L)
(2) calculate the amount of moles (using the molarity equation)
It is important to arrange the conversions in a way that allows for the cancellation of units. The final answer should have 3 sig figs.
125.0 mL NaOH 1 L
--------------------------- x --------------------- = 0.1250 L NaOH
1,000 mL
Molarity = moles / volume (L) <----- Molarity equation
0.190 M = moles / 0.1250 L <----- Insert values
0.0238 = moles <----- Multiply both sides by 0.1250
A solution of NaCl(aq) is added slowly to a solution of lead nitrate, Pb(NO3)2(aq), until no further precipitation occurs. The precipitate is collected by filtration, dried, and weighed. A total of 16.83g PbCl2(s) is obtained from 200.0mL of the original solution. Calculate the molarity of the Pb(NO3)2(aq) solution.
Answers
0.25 Molarity of lead nitrate aqueous solution is there when a solution of nacl is added slowly to a solution of lead nitrate.
What is molarity and how it is calculated out to be so?Molarity is the number of moles of substance present in one litre of solution and is measured in g/litres.Here in this question is given 16.83 g of lead nitrate is obtained from 200 ml of original solution.To calculate the molarity first we will have to calculate the number of moles of lead nitrate for which the formula is given mass/ molar mass.The mass that is given is 16.83 g and the molar mass is 331.2 g dividing we will get 5.02.Then dividing the number of moles by 0.2 L of solution we will get the answer as 0.25 g/litre molarity as the answer.To know more about molarity visit:
https://brainly.com/question/8732513
#SPJ10
How many molecules are in 24 grams of ozone (03)
Answers
Answer:48
Explanation:
Answer: 3. 0.125 X 10”23 molecules
Explanation:
Is a salamander a arthropod?
Answers
Answer:
no they are an theropod
Explanation:
I'm not completely sure but i hope this helps you maybe! :)
.Answer: No Because of their slender bodies and long tails, salamanders look somewhat like lizards, and people often confuse the two. But lizards are reptiles, whereas salamanders are amphibians (as are frogs and toads)they look really cool to play wit but no not 4 me.
Explanation:

Which of the following technique is used to purify the impurities that are not very different in chemical properties of element? [a] Gas chromatography [b] Column chromatography [c] TLC [d] HPLC
Answers
Answer:
Explanation: Liquid Chromatography
I'm sorry if i'm wrong
By referring to particles, explain why the white ring formed in the glass tube
Answers
Answer:
Ammonia and Hydrogen Chloride.
Explanation:
Assuming this is what you're referring to, Ammonia (NH3) and Hydrogen Chloride(NCI3). The concentrated ammonia (NH3) is placed on a pad in one end of a tube and hydrochloric acid (NCI3) on a pad at the other. Shortly after the gases will begin diffusing far enough to meet, a ring of solid ammonium chloride (NH4Cl) will be formed.
which element is found in period 6 group 14
Answers
Answer:
Lead
that can be found in the periodic table of elements
Explanation:
Water can be decomposed into its constituent elements, 2 H2O(l) → 2 H2(g) + O2(g), with an electrical current in a process called electrolysis. If 1.2 g of H2O is subjected to electrolysis and the products are collected in a cylinder with a volume of 0.20 L and a temperature of 30 °C, what is the partial pressure of H2?
Answers
The number of moles of hydrogen gas produced from 1.2 g of water is 0.06 moles. Then, the partial pressure of hydrogen gas is 4.9 atm.
What is partial pressure ?The partial pressure of a gas in a mixture is the contribution of the gas to the total pressure of the mixture.
The molar mass of water = 18 g/mol
mass = 1.2 g
number of moles = 1.2/18 =0.06 moles.
now the total pressure p = nRT/v
V = 0.20 L
T = 30°C =303 K
then p = (0.06 mol × 0.082 L atm/ K mol × 303 K) / 0.20 L = 7.4 atm.
Now, 2 moles of water gives 2 moles of hydrogen gas and one mole of oxygen. Then 0.06 moles of water gives 0.06 moles of hydrogen and 0.03 moles of oxygen.
Then, mole fraction of hydrogen = 0.06/(0.03 + 0.03) = 0.66.
Partial pressure of hydrogen = total pressure × mole fraction
= 0.66 × 7.4 atm = 4.9 atm.
Therefore, the partial pressure of hydrogen gas is 4.9 atm.
Find more on partial pressure :
https://brainly.com/question/13199169
#SPJ1
Balance and rewrite the following equation:
C₂H₂ + O2-----> CO₂ + H₂O
Answers
Ethyne gas undergo combustion reaction to give two moles of carbon dioxide and water as per the balanced equation written below:
\(\rm C_{2} H_{2} + \frac{5}{2} O_{2} \rightarrow 2CO_{2} + H_{2}O\)
What is combustion?Combustion is a type of reaction in which a gas burns in oxygen to give water and carbon dioxide. Hydrocarbons alkanes, alkenes or alkynes easily undergo combustion reaction and they can be used as fuels.
C₂H₂ is an alkyne names ethyne and it is an unsaturated hydrocarbon with triple bond between two carbon atoms. Ethyne gas reacts with oxygen to give two moles of carbon dioxide and one mole of water.
To balance the number of carbons the right side carbon dioxide is multiplied by 2 and the number of oxygens is balanced accordingly to get the balanced equation as follows:
\(\rm C_{2} H_{2} + \frac{5}{2} O_{2} \rightarrow 2CO_{2} + H_{2}O\)
To find more on combustion, refer here:
https://brainly.com/question/13153771
#SPJ1
From its position in the periodic table, determine which atom in each pair is more electronegative: (a) N or P (b) N or Ge (c) S or F (d) Cl or S (e) H or C (f) Se or P (g) C or Si
Answers
Answer:
p, get, s, s, c, se, SIse hhgggggggg I no
Answer:
1) Cl
2) O
3) O
4) S
5) N
6) P
7)N
Explanation:
7. What is the volume of the
composite
solid?
4 in.
3 in.
3 in.

Answers
Answer:
The volume of Component 1 is 36 cubic inches.
Explanation:
To calculate the volume of a composite solid, we need to determine the individual volumes of the different components and then add them together.
In this case, the composite solid consists of multiple components with the following dimensions:
Component 1:
Length: 4 inches
Width: 3 inches
Height: 3 inches
To find the volume of Component 1, we multiply the length, width, and height together:
Volume of Component 1 = Length x Width x Height = 4 in x 3 in x 3 in = 36 cubic inches
Therefore, the volume of Component 1 is 36 cubic inches.
Please provide the dimensions of the remaining components of the composite solid, and I will calculate the total volume by summing up the individual volumes.
What is the difference between structure and function in plants and animal cell organelles
Answers
Answer:
cell wall
Explanation:
plant cell have cell wall and can go through the process of plasmolysis.
they also contain stoma
animal cell are smaller
URGENT/
Drag the tiles to the correct locations. Each tile can be used more than once, but not all tiles will be used. Some locations will remain empty.
Chloramine has the chemical formula NH2Cl. Nitrogen has five valence electrons, each hydrogen has one valence electron, and chlorine has seven valence electrons. Complete the Lewis structure for this covalent compound.

Answers
The lewis structure of Chloamine can be given as above.
What is Covalent Compound?If one element shares electron with another elements to form bond. This is called as covalent bond. The compound which contain Covalent bond called Covalent compound.
What is Nitrogen atom?Nitrogen is one of the elements of periodic table. It has 7 as atomic number and 14 as mass number. Its electronic configuration is 1s2 2s2 2p3. It belongs to 15th group and 2nd period of the periodic table. It accept three electron to get stable electronic configuration.
As we know that, chloramine has the chemical formula NH2Cl. Nitrogen has five valence electrons, each hydrogen has one valence electron, and chlorine has seven valence electrons.
Thus, we concluded that Nitrogen form three single bond two with hydrogen and one with chlorine atom.
learn more about lewis structure:
https://brainly.com/question/20300458
#SPJ1

Drag the tiles to the correct boxes to complete the pairs. Not all tiles will be used.
Match each SI unit to the quantity it measures.
Answers
The SI unit to the quantity it measures are:
mass - kilogram, gramtemperature - kelvintime - second, nanosecondelectric current - ampereWhat is SI unit used for?Mass: The mass of an object is a measure of its amount of matter. The SI unit of mass is the kilogram (kg) or gram (g).
Temperature: Temperature is a measure of the average kinetic energy of the particles in a substance. The SI unit of temperature is the kelvin (K).
Time: Time is a measure of the interval between two events. The SI unit of time is the second (s).
Electric current: Electric current is a measure of the flow of electric charge. The SI unit of electric current is the ampere (A).
Find out more on SI unit here: https://brainly.com/question/16393390
#SPJ1
Complete question:
Drag the tiles to the correct boxes to complete the pairs. Not all tiles will be used.
Match each SI unit to the quantity it measures.

An unidentified gas is determined to be 24.0% carbon and 76% fluorine by mass. What is the empirical formula of this gas
Answers
Answer:
CF₂
Explanation:
Let's assume we have 100 g of the gas. If that were the case we'd have
24 g of C76 g of FNow we convert both masses into moles, using their respective molar mass:
24 g C ÷ 12 g/mol = 2 mol C76 g F ÷ 19 g/mol = 4 mol FWe can express those results as C₂F₄.
To determine the empirical formula we reduce those coefficients to the lowest possible integers, leaving us with CF₂.
which statement describes virginia's watershed i need this ASAP pls help
Answers
B: It has fewer than 10 total watersheds best describes
What is a watershed?A watershed is the area of land where all of the water that falls in it and drains off of it goes to a common outlet. All the land drains water into rivers that drain into the Chesapeake Bay, where it enters the Atlantic Ocean.
Why is it called a watershed?The area that drains into a single river is the watershed for that river.
Learn more about Watershed here
https://brainly.com/question/24813719
#SPJ2
What ion has a +3 charge, 28 electrons and an atomic mass of 71?
Answers
The ion with a +3 charge, 28 electrons, and an atomic mass of 71 is the aluminum ion (\(Al^{3+}\)).
Aluminum (Al) typically has an atomic number of 13, which means it has 13 protons and 13 electrons in its neutral state. However, in the given ion, \(Al^{3+}\), the ion has lost three electrons, resulting in a +3 charge. This means that the ion now has 13 protons and only 10 electrons remaining, giving it a net positive charge of +3.
The atomic mass of aluminum is 26.98 atomic mass units (amu). The given ion has an atomic mass of 71 amu, which suggests that the ion has gained additional particles. In this case, the ion has also gained three neutrons, resulting in a higher atomic mass.
The total number of particles (protons, neutrons, and electrons) in the ion can be calculated by adding the number of protons (13) and the number of neutrons (3), which equals 16. Since the ion has a net charge of +3, it only contains 10 electrons.
In summary, the ion with a +3 charge, 28 electrons, and an atomic mass of 71 is the aluminum ion (\(Al^{3+}\)), which has 13 protons, 10 electrons, and 3 neutrons.
for such more questions on electrons
https://brainly.com/question/26084288
#SPJ8
A pharmacist quizzes a pharmacy intern on the aliquot method in the preparation of 12 capsules each to contain 80 mg of morphine sulfate and 3.2 mg of naltrexone hydrochloride. Lactose is to be used as a diluent, a prescription balance with a sensitivity of 6 mg is proposed, and a 4% error is acceptable. Provide the relevant calculations.
Answers
To prepare 12 capsules each containing 80 mg of morphine sulfate and 3.2 mg of naltrexone hydrochloride, the following calculations can be used:
Calculate the total weight of the morphine sulfate and naltrexone hydrochloride: 80 mg + 3.2 mg = 83.2 mg
Calculate the weight of the diluent required: 12 capsules * 83.2 mg/capsule = 999.6 mg
Calculate the volume of the diluent required based on its density (assume a density of 0.9 g/mL for lactose): 999.6 mg / (0.9 g/mL) = 1.11 mL
Calculate the weight of the diluent required based on its volume: 1.11 mL * 0.9 g/mL = 1 g
Calculate the amount of the diluent required based on the sensitivity of the prescription balance: 1 g / (6 mg/g) = 166.7 mg
The amount of the diluent required according to this calculation is 166.7 mg. This amount should be rounded up to the nearest whole number (167 mg) to account for the 4% error tolerance. The final aliquot would therefore contain 167 mg of lactose, 80 mg of morphine sulfate, and 3.2 mg of naltrexone hydrochloride.
help me pleaseeeeee

Answers
Answer:
Arthropod (3)
Annelids (2)
Cnidarians (1)
Echinoderms (4)
Explanations:
I hope all this helps if u don't understand it let me know.
What compound are formed with lithium and selenium?
Answers
Answer:
Lithium selenide Lithium selenide (Li2Se) 12136-60-6 Dilithium selenide lithium selenidolithium
Molecular Weight 92.9 g/mol
Dates Modify 2020-11-15 Create 2005-08-08
Answer:
Lithium is +1 and Selenium is -2, so Li2Se (Lithium Selenide).
benzen has a boiling point of 80.10 c we know the change in boiling point for a solution of c6h14 in benzen is 2.25 what is the new boiling point for the solution
bp= ? c
Answers
The pressure of the environment affects the liquid's boiling point. The boiling point of the liquid is higher when it is under high pressure than when it is under normal atmospheric pressure. For a given pressure, various liquids have different boiling points.
The temperature at which a liquid's vapour pressure equals the surrounding atmosphere is known as the boiling point of the liquid. This temperature causes the liquid to become a vapour.
The temperature of the liquid, the pressure of the atmosphere, and the pressure of the vapour all affect its boiling point.
We know that change in temperature of a system is given by the following formula:
Initial boiling point (T₁) = 80.1 °C
ΔT = T₂ - T₁
2.25 = T₂ - 80.1
T₂ = 82.35 °C
To know more about boiling point, visit;
https://brainly.com/question/28986258
#SPJ1
Answer:
82.35
Explanation:
acellus
For alkyl halides used in SN1 and SN2 mechanisms, rank the leaving groups in order of reaction rate. You are currently in a ranking module. Turn off browse mode or quick nav, Tab to move, Space or Enter to pick up, Tab to move items between bins, Arrow Keys to change the order of items, Space or Enter to drop.
Answers
Answer:
Iodide> Bromide > chloride > flouride
Explanation:
During a nucleophilic substitution reaction, a nucleophilie replaces another in a molecule.
This process may occur via an ionic mechanism (SN1) or via a concerted mechanism (SN2).
In either case, the ease of departure of the leaving group is determined by the nature of the C-X bond. The stronger the C-X bond, the worse the leaving group will be in nucleophilic substitution. The order of strength of C-X bond is F>Cl>Br>I.
Hence, iodine displays the weakest C-X bond strength and it is thus, a very good leaving group in nucleophillic substitution while fluorine displays a very high C-X bond strength hence it is a bad leaving group in nucleophilic substitution.
Therefore, the ease of the use of halide ions as leaving groups follows the trend; Iodide> Bromide > chloride > flouride
What is a single called organism able to do
Answers
Answer:
Single-celled organisms are able to carry out all the processes of life without help from other cells. Multicellular organisms carry out their life processes through division of labor. They have specialized cells that do specific jobs.
Explanation:
that might be the answer
Answer:
All single-celled organisms contain everything they need to survive within their one cell. These cells are able to get energy from complex molecules, to move, and to sense their environment. The ability to perform these and other functions is part of their organization.
Hope it helps
Explanation:
Ice has a density of 0.92g/cm3. It will float in water.
Answers
The density of pure water is 1 g/cm^3.
Its density is 0.98 g cm 3 at room temperature, in comparison with the handiest zero.92 g cm 3 for ice, a reality that has to be defined through atomic, and molecular concepts. If ice has been no longer much less dense than water, it might sink, having a devastating impact on lake backside ecosystems. believe it or now not, ice is honestly about 9% much less dense than water. for the reason that water is heavier, it displaces the lighter ice, causing the ice to glide to the pinnacle.
The density of ice is about 90 percent that of water, but that could range because ice can contain air, too. meaning that about 10 percent of an ice cube or iceberg will be above the water line. The density of water is maximum at four∘C, and the density of the ice is much less than the water due to its susceptible intermolecular pressure of attraction. as the density of water is more, it's miles heavier than ice. therefore ice floats on the floor of the water. Ice continually floats due to the fact it's far less dense than everyday water. because frozen water molecules shape a crystal.
Learn more about A density here:-https://brainly.com/question/17780219
#SPJ9
1. In the above reaction, the reactants are
and
Answers
Answer:
PLZZ like it tuk me to long to explan
Explanation:
The reaction between zinc and sulfur can be shown in what is called a chemical equation . In words, we could write the reaction as:
zinc + sulfur → zinc sulfide
The more convenient way to express a chemical reaction is to use the symbols and formulas of the substances involved:
Zn + S → ZnS
The substance(s) to the left of the arrow in a chemical equation are called reactants. A reactant is a substance that is present at the start of a chemical reaction. The substance(s) to the right of the arrow are called products . A product is a substance that is present at the end of a chemical reaction. In the equation above, the zinc and sulfur are the reactants that chemically combine to form the zinc sulfide product.
There is a standard way of writing chemical equations. The reactants are all written on the left-hand side of the equation, with the products on the right-hand side. An arrow points from the reactants to the products to indicate the direction of the reaction:
reactants → products
When appropriate, a symbol may be written above or below the arrow to indicate some special circumstance. The symbol “Δ” is often used to indicate that the reaction is to be heated.
The presence of the arrow also indicates that the reaction goes in one direction under the conditions indicated. There are reactions which can be easily reversed, but we will not take those up right now.
There are a wide variety of reactions possible: elements may form compounds (as seen in the reaction above), compounds may form elements (water will break down in the presence of an electric current to form hydrogen gas and oxygen gas) or compounds may combine, break apart, or rearrange to form new materials.
How is this compound classified C4H6O4
Answers
The compound C4H6O4 can be classified as a dicarboxylic acid. In this case, the presence of four carbon atoms (C4) indicates that it is a relatively larger molecule.
The molecular formula also contains six hydrogen atoms (H6) and four oxygen atoms (O4). The presence of oxygen and carbon atoms suggests the possibility of carboxyl groups (-COOH) in the compound. Carboxyl groups are functional groups consisting of a carbonyl group (C=O) and a hydroxyl group (-OH) attached to the same carbon atom.
Since the compound contains four oxygen atoms, it is possible that it contains two carboxyl groups. A compound with two carboxyl groups is classified as a dicarboxylic acid. Dicarboxylic acids are organic compounds that have two carboxyl functional groups.
They are characterized by their ability to donate two protons (H+) and act as acids. Therefore, based on the molecular formula C4H6O4, the compound is classified as a dicarboxylic acid.
For more such questions on compound
https://brainly.com/question/15929599
#SPJ11
a mountain
b trench
c mid ocean range
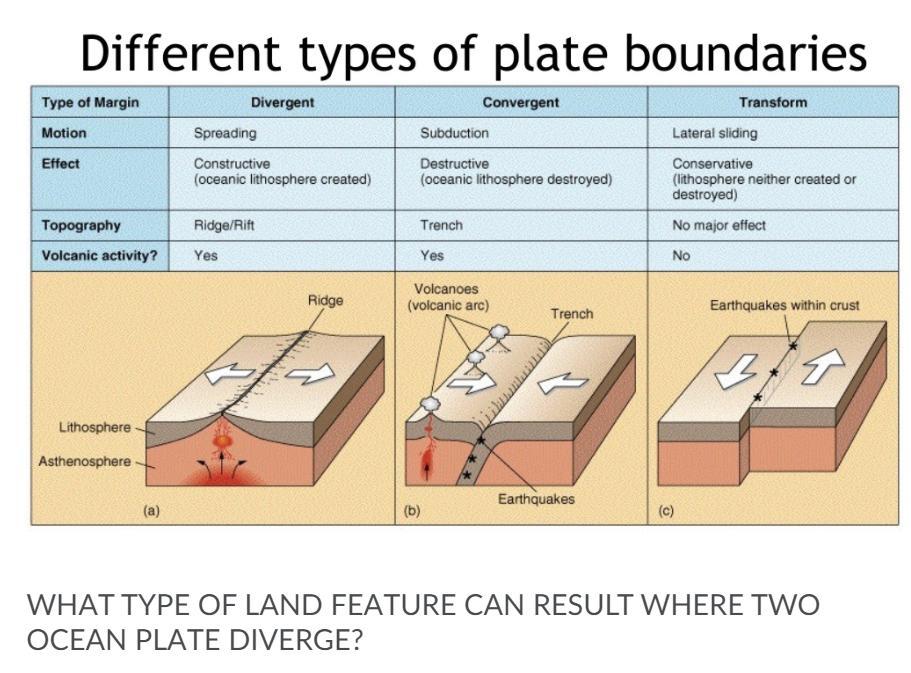
Answers
how do you determine if a bond is ionic or covalent
