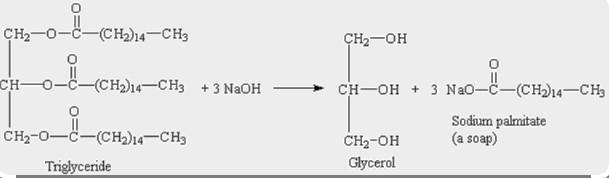
1) write a balanced equation to show the hydrolysis of glycerol tristearate (tristearin, a triple ester of glycerol) using water and sodium hydroxide to make soap. in addition to the molar ratios, give the mass ratios, i.e., the ratios of masses that react according to the reaction stoichiometry) based on 20 g of tristearin.
Answers
Mass ratio of Tristearin that react according to the reaction : \(NaOH\) is
20 : 27
The main fat in beef is tristearin. A molecule of glycerine that has interacted with three (3) molecules of the fatty acid stearic acid is known as a triglyceride.
1 mole of Tristearin requires 3 moles of \(NaOH\) to react to give 1 mole of glycerol & 3 moles of sodium sterate.
Molar ratio of reactant Tristearin : \(NaOH\) = 1 : 3
Molar weight of Tristearin = 891.5 g/mol
=> 20 g Tristearin = \(\frac{20}{891.5} moles\)
=> \(NaOH\) required to react with \(\frac{20}{891.5}\) moles of Tristearin = \(3\times\frac{20}{891.5} moles\) = 0.0673 moles
=> Molecular weight of \(NaOH\) = 40 g/mol
=> \(NaOH\) mass required to react with 20 g Tristearin = 0.0673 × 40g = 2.7g
∴ Mass ratio of Tristearin: \(NaOH\) is 20 : 27.
Learn more about Tristearin:
brainly.com/question/7175814
#SPJ4
Related Questions
1. Write the IUPAC names for the following 1.1 1.2 N 1.3 O NO2 x Y ·0 OH 5
Answers
1. The IUPAC name of N is nitrogen.
2. Nitrogen dioxide
3.The IUPAC name of O is oxygen
4.The IUPAC name of OH is hydroxyl.
The IUPAC name of ·0 is a radical. It is commonly found in organic chemistry and plays an important role in many reactions.
IUPAC names for the given compounds are:1.1. N: Nitrogen
The IUPAC name of N is nitrogen.
It is a non-metal and belongs to group 15 in the periodic table. It has an electronic configuration of 1s2 2s2 2p3.1.2. NO2: Nitrogen dioxide
Explanation: NO2 is a chemical compound that is formed by the combination of nitrogen and oxygen. It is a reddish-brown gas that has a pungent odor.
The IUPAC name of NO2 is nitrogen dioxide.1.3. O: Oxygen
Explanation: The IUPAC name of O is oxygen.
It is a non-metal and belongs to group 16 in the periodic table. It has an electronic configuration of 1s2 2s2 2p4.
X: UnknownExplanation: No IUPAC name can be given to an unknown compound as the structure and composition are not known.
Y: Hydroxyl Explanation: The IUPAC name of OH is hydroxyl.
It is a functional group that is composed of an oxygen atom and a hydrogen atom (-OH). It is commonly found in alcohols and phenols. ·0: RadicalExplanation: A radical is a molecule or an ion that contains an unpaired electron.
for more question on electronic configuration
https://brainly.com/question/26084288
#SPJ8
Note: The complete question is given below
Provide the IUPAC names for the following compounds:
\(CH_3CH_2CH(CH_3)CH_2CH_2CH_2CH_3\)
C6H5CH(CH3)2
H2NCH2CH2CH2CH2CH2NH2
CH3CH2CH2CH2CH2OH
CH3CH2CH2CHOHCH3
A cold air mass and a warm air mass are predicted to meet. What type of weather can be expected between the two air masses?
A.
warm weather
B.
cold weather
C.
dry weather
D.
stormy weather
Answers
Answer:
Explanation:
C. warm weather
Answer:
stormy weather
Convert 100.6 Kelvin to degrees C.
°C = K - 273
[?] °C
Answers
Answer:
-172.6 °C
Explanation:
You want to know the Celsius equivalent of the temperature 100.6 K.
ConversionThe relation is ...
C = K - 273.15
C = 100.6 -273.15 = -172.55
The temperature is -172.55 °C, about -172.6 °C.
__
Additional comment
We have rounded to tenths, because that is precision of the temperature given. If you use 273 as the conversion constant, you will get -172.4.
Which statement best describes the temperature of the region at the equator?
Answers
The statement that should describe the temperature of the region at the equator is that it can be associated with perpendicular solar radiation.
What is the Equator line?The Equator line at zero (0) latitude is an imaginary line that divides the Earth's planet into two equal parts composed of South latitudes and North latitudes.
Temperatures at low latitudes, i.e., near the Equator line, are warmer because this region receives perpendicular solar rays all the year, thereby the energy of solar radiation is distributed to the Earth's surface.
In conclusion, the statement that should describe the temperature of the region at the equator is that it can be associated with perpendicular solar radiation.
Learn more about the Equator line here:
https://brainly.com/question/1264608
#SPJ1
A substance has a density of 13.5 g/mL.
You have a 30.0g sample. What is the
volume of your sample?
Answers
The volume of your sample is when a substance has a density of 13.5 g / mL with mass 30.0 g .
The density of a place refers to the quantity of things—which may include people, animals , plants, or objects—there are in it. The density can determine by just divide the mass of objects by the volume measurement .
Example : A country's population density is calculated by dividing its total population by its area, measured in square kilometers or miles.
Formula used :
d = m / v ( 1 )
where d = Density of the sample = 13.5 g/mL.
m = mass = 30.0 g
V = volume = ?
Put the value of mass m and density in equation ( 1 ) we get ,
13.5 g/mL = 30.0 g / volume
volume = 30.0 g / 13.5 g/mL = 2.22 ml
to learn more about Density
https://brainly.com/question/15164682
#SPJ1
How many grams of NaCl

Answers
You would recover 36.525g of NaCl after evaporating all of the water.
How to find the how many grams of NaCl that would be recover when all water is evaporated off of this solution?To find the grams of NaCl that would be recovered after evaporating all the water, we can use the following formula:
mass = moles * molar mass
Where:
Moles = Molarity * Volume
Molarity = 0.250 M
Volume = 2500.0 mL = 2.5 L
Molar mass of NaCl = 58.44 g/mol
mass = 0.250 M * 2.5 L * 58.44 g/mol
mass = 36.525 g
Learn about evaporation here https://brainly.com/question/2013258
#SPJ1
An element has 2 stable isotopes. One has 13 amu and 1.07% abundant . The second has 12 amu and 98.93% abundant. What is the average atomic mass of the element
Answers
The average atomic mass of the element is 12.0107 amu.
To calculate the average atomic mass of the element in question, we can use the following formula:
average atomic mass = (mass of isotope 1 x abundance of isotope 1) + (mass of isotope 2 x abundance of isotope 2)
where "mass of isotope 1" is the mass of the first stable isotope (13 amu in this case), "abundance of isotope 1" is the percentage of that isotope in the element (1.07% in this case), "mass of isotope 2" is the mass of the second stable isotope (12 amu in this case), and "abundance of isotope 2" is the percentage of that isotope in the element (98.93% in this case).
Substituting the given values in the formula, we get:
average atomic mass = (13 amu x 1.07%) + (12 amu x 98.93%)
average atomic mass = (0.1391 amu) + (11.8716 amu)
average atomic mass = 12.0107 amu
Therefore, the average atomic mass of the element is 12.0107 amu.
This means that on average, one atom of this element weighs 12.0107 atomic mass units (amu), which is slightly heavier than the most abundant isotope (12 amu) due to the presence of the less abundant isotope (13 amu). This concept is important in chemistry because the mass of atoms plays a crucial role in determining their chemical and physical properties. The knowledge of the average atomic mass of an element is important in a wide range of applications, including analytical chemistry, geochemistry, and nuclear physics.
Know more about atomic mass here:
https://brainly.com/question/3187640
#SPJ11
An atomic cation with a charge of +1 has the following electron configuration:
1s 2s 2p 3s 3p 3d ¹4s¹
What is the chemical symbol for the ion?
How many electrons does the ion have?
How many 3p electrons are in the ion?
Answers
A) The chemical symbol for the ion is Fe+
B) It has 20 electrons in total, and there are 6 3p electrons in the ion.
C) There are 6 electrons present in the 3p orbital.
The atomic cation with the given electron configuration is represented by the chemical symbol Fe+.
To determine the number of electrons in the ion, we need to count the electrons present in the electron configuration. In the given configuration, we can see that the 1s orbital has 2 electrons, the 2s orbital has 2 electrons, the 2p orbital has 6 electrons, the 3s orbital has 2 electrons, the 3p orbital has 6 electrons, the 3d orbital has 1 electron, and the 4s orbital has 1 electron. Adding up these numbers, we have:
2 + 2 + 6 + 2 + 6 + 1 + 1 = 20
Therefore, the ion has 20 electrons.
To determine the number of 3p electrons in the ion, we look at the 3p orbital. In this case, there are 6 electrons present in the 3p orbital.
In summary, the chemical symbol for the ion is Fe+, it has 20 electrons in total, and there are 6 3p electrons in the ion.
For more question on electrons
https://brainly.com/question/26084288
#SPJ8
Aluminum bromide can be prepared by reacting small pieces of aluminum foil with liquid bromine at room temperature. The balanced chemical reaction is:2Al(s) + 3Br2(l) → 2AlBr3(s)How many moles of Br2 are needed to produce 5 mol of AlBr3, if sufficient Al is present?
Answers
Based on the mole ratio from the chemical reaction 3 moles of Br2 produces 2 moles of AlBr3. We can set this equation up to determine the unknown:
\(\begin{gathered} \frac{2}{3}=\frac{x}{5} \\ 3x=2\times5 \\ x=\frac{10}{3} \\ x=3.3moles \end{gathered}\)3.3 moles of Br2 is needed to reacts with 5 mol of AlBr3.
Identify which of these molecules has the highest boiling point and give the reasoning why in terms of intermolecular force. CHCl3, H2O, CO2
Answers
According to the forces of attraction, water has highest boiling point as it possesses hydrogen bonding.
What are forces of attraction?Forces of attraction is a force by which atoms in a molecule combine. it is basically an attractive force in nature. It can act between an ion and an atom as well.It varies for different states of matter that is solids, liquids and gases.
The forces of attraction are maximum in solids as the molecules present in solid are tightly held while it is minimum in gases as the molecules are far apart . The forces of attraction in liquids is intermediate of solids and gases.
The physical properties such as melting point, boiling point, density are all dependent on forces of attraction which exists in the substances.
Learn more about forces of attraction,here:
https://brainly.com/question/23841038
#SPJ1
m A 10.00g sample of a substance is found to contain 5.12g of water. What is the percent by of water in the compound? A
. 5.12% B. 4.88% C. 48.8% D. 51.2%
Answers
Answer:
The answer is D) 51.2%
Explanation:
5.12g of 10.00g of the substance is water:
percentage of water= 5.12 x 10.00 =51.2%
Answer:
b
Explanation:
A metal carbonate, XCO3 of mass 2.012 g was heated resulting in the formation of XO, a metal oxide and carbon dioxide with a mass of 0.855 g according to the reaction shown below: XCO3 (s) → XO (s) + CO2 (g) (Atomic mass of O-15.999 g/mol; H-1.008 g/mol; C-12.011 g/mol).
Answers
The metal X has an approximate molar mass of 42.36 g/mol and the metal is most likely calcium.
What is the molar mass of XCO₃?The molar mass of the metal carbonate XCO₃ and identify the metal X, we need to calculate the number of moles of XCO₃ and CO₂ using the given masses and molar masses.
The molar mass of CO₂ (carbon dioxide) is 12.011 g/mol (for carbon) + 2 * 15.999 g/mol (for oxygen) = 44.01 g/mol.
The number of moles of CO₂ can be calculated using the formula:
moles of CO₂ = mass of CO₂ / molar mass of CO₂
moles of CO₂ = 0.855 g / 44.01 g/mol
moles of CO₂ ≈ 0.01944 mol
Since the reaction stoichiometry is 1:1 between XCO₃ and CO₂, the number of moles of XCO₃ is also approximately 0.01944 mol.
molar mass of XCO₃ = mass of XCO₃ / moles of XCO₃
molar mass of XCO₃ = 2.012 g / 0.01944 mol
molar mass of XCO₃ ≈ 103.38 g/mol
The molar mass of XCO₃ is approximately 103.38 g/mol.
To determine the metal X:
molar mass of X = molar mass of XCO3 - molar mass of CO3
molar mass of X = 103.38 g/mol - (12.011 g/mol + 3 * 15.999 g/mol)
molar mass of X ≈ 42.36 g/mol
Metal X is most likely Calcium that has a molar mass of 40 g/mol
Learn more about molar mass at: https://brainly.com/question/837939
#SPJ1
What is the pH of a 3.5 x 10-5 M solution of HCl?
Answers
Answer:
pH = 4.45
Explanation:
We need to find the pH of \(3.5\times 10^{-5}\ M\) solution of HCl. We know that, pH of a solution is given by :
\(pH=-log[H]^+\)
Put all the values,
\(pH=-log[3.5\times 10^{-5}]\\\\pH=4.45\)
So, the pH of the solution of HCl is 4.45.
Find five household items that contain ionic compounds. List the following information for each compound: a.name of the product
b. the chemical name of the ionic compound
d.the chemical formula of the ionic compound c. identify the ions present in each compound
Answers
Answer:
D . GOD BLESS .
Explanation:
Muối sunfua với hidro sunfua có giống nhau về tính chất vật lý không ạ. Ứng dụng và điều chế muối sufua như thế nào ạ
Answers
the answer is is is is is is is is is is is is is is
Question Which type of object creates the most friction? Responses hard objects hard objects rough objects rough objects smooth objects smooth objects soft objects
Answers
The type of object that creates the most friction is a rough object.
What is frictional force?This is the force that opposes the motion of an object. It usually exist between two surfaces that are in contact.
Factors affecting frictional force include the following;
The frictional force between two bodies depends mainly on the following factors:
(I) forces acting on the objects
(ii) type of the surface in contact
(iii) position or inclination of the surfaces
How to reduce frictional force between two surfaces;
Frictional force can be reduced with lubricantsFrictional force can be reduced using bearings in moving parts,Frictional force can reduced using inclined surfacesThus, the type of object that creates the most friction is a rough object.
Learn more about frictional force here: https://brainly.com/question/4618599
#SPJ1
How has the increase in human population impacted earths resources?
Answers
Answer:
The increase in human population has impacted Earth's resources in several major ways:
• Increased demand for food, water, and shelter. A larger population requires massive increases in food, water, and living spaces which strains natural resources and infrastructure. Producing enough food alone is a significant challenge.
• Accelerated consumption of resources. As population grows, the use of resources like forests, minerals, fossil fuels also increases rapidly to meet demands. This accelerated depletion of resources threatens long term sustainability.
• Increased pollution. A bigger population produces more pollution, waste, emissions, and environmental degradation as a byproduct of energy usage, transportation, industrialization, and land/resource use. This pollution harms ecosystems and contaminates the air, water and land.
• Biodiversity loss. As natural habitats are destroyed or fragmented to enable more human use, many plant and animal species lose their homes and face a higher risk of extinction. Tropical rainforests, in particular, have been heavily impacted.
• Inequality. While resources are limited, population growth often exacerbates inequality in access to and distribution of resources. Poor or developing regions typically have the highest populations but fewest resources per capita.
• Migration and conflict. Shortages of resources in certain areas or regions may lead to migration, economic troubles, social unrest, and in some cases even resource conflicts or wars.
• Slower development. Extremely rapid population growth rates make it difficult for governments, organizations and societies to effectively manage development, improve standards of living, advance technology, and make other progress at an optimal pace. Slower, more stabilized population growth may enable a higher overall quality of life.
So in many profound and troubling ways, increased population size has created immense pressures on Earth's resources and made it more difficult to meet present and future needs in a sustainable manner. Most experts agree that slowing population growth is critical to ensuring resource security for future generations.
Explanation:
how many mL of 0.100M NaCl would be required to make a 0.0350M solution of NaCl when diluted to 150.0mL with water?
Answers
The volume of 0.100M NaCl that would be required to make a 0.0350M solution of NaCl when diluted to 150.0mL with water is 52.5mL.
How to calculate volume?The volume of a diluted solution can be calculated using the following formula;
CaVa = CbVb
Where;
Ca = initial concentrationVa = initial volumeCb = final concentrationVb = final volumeAccording to this question, 0.100M NaCl would be required to make a 0.0350M solution of NaCl when diluted to 150.0mL with water. The volume can be calculated as follows:
0.1 × Va = 0.0350 × 150
0.1Va = 5.25
Va = 52.5mL
Therefore, 52.5mL is the volume required to dilute the sodium chloride solution.
Learn more about volume at: https://brainly.com/question/22762236
#SPJ1
Does the sodium-potassium pump go to work prior to or after the contraction has occurred? Explain your response.
(NEED HELP ASAP. THANK U FOR UR HELP)
Answers
The sodium-potassium pump goes to work after the contraction has occurred in order to repolarize the cells.
What is the sodium-potassium pump?The sodium-potassium pump is a pump that functions in transporting sodium ions out of the cell and potassium ions inside the cell.
At the resting membrane potential, the sodium-potassium pump ensures that the interior of the cell is negative relative to outside the cell because three sodium ions are pumped out of the cell for every two potassium ions that are pumped into the cell.
When an action potential reaches the cell, the pump opens and allows sodium ions into the cell thereby depolarizing it. Afterward, the sodium-potassium pump works to repolarize the cell by pumping sodium out and potassium in.
Learn more about sodium-potassium pump at: https://brainly.com/question/11673875
#SPJ1
modern commercial airlines are largely made of aluminum, and light and strong metal. But the fact that aluminum is cheap enough that airplanes can be made out of it is a bit of historical luck. before the discovery of the Hall-Heroult process in 1886, aluminum was as rare and expensive as gold. What would happen if airplanes had to be made of steel? The fuselage of a Boeing 787, which can carry 400 passengers, is approximately a hollow aluminum cylinder without ends, 70.7 m long, 6.5 m wide, and 2.5 mm thick. Suppose this fuselage was made of steel (density 7.87 g/cm^{3}) instead of aluminum (density 2.7g/\(cm^{3}), and let's say the average passenger has a mass of 81 kg. We'll also assume the engines can't lift any greater mass than they already do. Calculate the number of passengers that the Boeing 747 could carry if its fuselage was made of steel.
Answers
Answer:
170 passenger . ( approx )
Explanation:
Total volume of metal making fuselage = π ( r₂² - r₁² ) l
r₂ is outer radius of cylindrical tube like fuselage
= 6.5 / 2 + .0025 = 3.2525 m
r₁ is inner radius of cylindrical tube like fuselage
= 6.5 / 2 = 3.25 m
l = 70.7 m
Total volume of fuselage = π ( r₂² - r₁² ) l
= 3.14 x 70.7 x ( 3.2525² - 3.25² )
= 221.998 ( 10.57875 - 10.5625)
= 3.60747 m³
If it is made of aluminium , its mass
= volume x density
= 3.60747 x 2700 kg
= 9740.16 kg
weight of passenger = 81 x 400 = 32400 kg
Total mass = 32400 + 9740.16
= 42140.16 kg
If it is made of iron , its mass
= volume x density
= 3.60747 x 7870 kg
= 28390.79 kg
If number of passenger required be n
81 n + 28390.79 = 42140.16 ( as per condition given )
n = 170 passenger . ( approx )
How many particles are there in 0.057 moles of lithium bromide made
Answers
There are 3.44 x 10^{22} particles in 0.057 moles of lithium bromide.
What chemical compound is lithium bromide known by?The lithium bromide formula also known as the lithium monobromide formula or Bromo lithium formula is explored. It is a counterion bromide-based salt of lithium.
we have to use Avogadro's constant,
Avogadro's constant, is approximately equal to 6.022 x 10^{22} particles per mole.
we can use the following formula:
number of particles = moles x Avogadro's constant
Substitute the values,
number of particles = 0.057 moles x 6.022 x 10^{23} particles/mol
Simplifying the equation
number of particles = 3.44 x 10^{22} particles
To know more about lithium bromide visit:
https://brainly.com/question/16584013
#SPJ9
weather balloon is filled with helium to a volume of 340 L at 30 ∘C and 751 mmHg . The balloon ascends to an altitude where the pressure is 495 mmHg and the temperature is -27 ∘C.What is the volume of the balloon at this altitude?
Answers
Answer: Thus the volume of the balloon at this altitude is 419 L
Explanation:
Combined gas law is the combination of Boyle's law, Charles's law and Gay-Lussac's law.
The combined gas equation is,
\(\frac{P_1V_1}{T_1}=\frac{P_2V_2}{T_2}\)
where,
\(P_1\) = initial pressure of gas = 751 mm Hg
\(P_2\) = final pressure of gas = 495 mm Hg
\(V_1\) = initial volume of gas = 340 L
\(V_2\) = final volume of gas = ?
\(T_1\) = initial temperature of gas = \(30^oC=273+30=303K\)
\(T_2\) = final temperature of gas = \(-27^oC=273-27=246K\)
Now put all the given values in the above equation, we get:
\(\frac{751\times 340}{303}=\frac{495\times V_2}{246}\)
\(V_2=419L\)
Thus the volume of the balloon at this altitude is 419 L
what is the Thermodynamics of leaching of ilmenite in hydrochloric acid for titania pigment production?
Answers
The thermodynamics of leaching of ilmenite in hydrochloric acid for titania pigment production is an industrial production of nano-sized titanium dioxide (TiO2).
What is thermodynamics?Thermodynamics is defined as the change in the energy of system that brings about work in that system.
The leaching of ilmenite which is a titanium-iron oxide mineral with HCl can lead to the production of titanium dioxide (TiO2).
The use of thermodynamics in this process controls the heat and energy of the leaching system leading to a better yield of final products.
Learn more about thermodynamics here:
https://brainly.com/question/13059309
#SPJ1
Which of the following is a possible way to describe the H2O component in the reaction below?
2HCI(aq) + Ca(OH)2(aq) --> 2H2O(I) + CaCl2(aq)

Answers
that H2O is on the opposite end of the equation, this is one of the reaction's products. We can therefore refer to the H2O element as just a product.
By a product, what do you mean?The thing being sold is called a product. An item or service can be considered a product. Physical, virtual, or cyberspace forms are all possible. Every good has a price because it costs money to produce it. The market, quality, marketing, and intended market group all affect the price which can be paid.
Describe by-product using an example.Anything that is created while another product is being manufactured or processed is known as a by-product. The by-product of is the source of the raw material.
To know more about product visit:
https://brainly.com/question/1851669
#SPJ1
Answer:
the correct ansrew is c
Explanation:
because there are 2 molecules of h2o
Classify the strongest type of intermolecular force in the following interactions: solvent-solvent, solvent-solute, and solute-solute when solid glucose (C6H12O6) is placed in the water. Drag the terms on the left to the appropriate blanks on the right to complete the sentences
hydrogen bonding
dipole-dipole forces
London dispersion forces
ion-dipole forces
Solute-solute is .
Solvent-solvent is .
Solvent-solute is .
please fill one sentence for each blank
Answers
The interaction between polar water molecules and the partially charged ions of glucose, resulting in ion-dipole forces, is the strongest inter molecular force in the solvent-solute interaction.
What is the solute interaction?Solute interaction is the interaction between two or more solutes in a solution. This interaction can be either physical or chemical in nature. Physical interactions involve the solutes having an effect on each other due to their shape, size or charge. Chemical interactions involve the solutes reacting with each other to form new compounds. In either case, the solutes will have an effect on the properties of the solution such as viscosity, boiling point, freezing point, and solubility. Solutes can also interact with each other when they are mixed together, as in a mixture of two liquids or a mixture of two solids.
To learn more about solute interaction
https://brainly.com/question/14805127
#SPJ1
gas syringe
bung
chips
25 cm of dilute
hydrochloric acid
Which channes slow down the rate of reaction?
Decrease the size of pieces of marble chips
Decrease surface area of marble chips
Inercase concentration of acid
Increase temperature of acid
Answers
Answer:
Decrease surface area of marble chips.
Explanation:
Because the reaction goes on the surface of marble chips, decreasing surface area of marble chips will decrease(slow down) the rate of the reaction.
HQ5.40
Homework Answered Due Today, 11:59 PM
The reaction 3H₂(g) + N₂(g) → 2NH3(g) has an enthalpy of reaction of -92.6 kJ/mol. If 1 g of hydrogen and 2 g of nitrogen are
reacted, how much heat is produced (kJ)?
Answers
The amount of heat energy produced when 1 g of hydrogen and 2 g of nitrogen are reacted, is -6.61 KJ
How do i determine the heat energy produced?First, we shall obtain the limiting reactant. Details below:
3H₂ + N₂ -> 2NH₃
Molar mass of N₂ = 28 g/molMass of N₂ from the balanced equation = 1 × 28 = 28 g Molar mass of H₂ = 2 g/molMass of H₂ from the balanced equation = 3 × 2 = 6 gFrom the balanced equation above,
28 g of N₂ reacted with 6 g of H₂
Therefore,
2 g of N₂ will react with = (2 × 6) / 28 = 0.43 g of H₂
We can see that only 0.43 g of H₂ is needed in the reaction.
Thus, the limiting reactant is N₂
Finally, we the amount of heat energy produced. Details below:
3H₂ + N₂ -> 2NH₃ ΔH = -92.6 KJ
Molar mass of N₂ = 28 g/molMass of N₂ from the balanced equation = 1 × 28 = 28 gFrom the balanced equation above,
When 28 grams of N₂ reacted, -92.6 KJ of heat energy were produced.
Therefore,
When 2 grams of N₂ will react to produce = (2 × -92.6) / 28 = -6.61 KJ
Thus the heat energy produced from the reaction is -6.61 KJ
Learn more about heat energy:
https://brainly.com/question/31429264
#SPJ1
Determine the
Cl
for NeMut1:Wt using the data presented in part 2 of the case study.
1×10 −10
1×10 10
1×10 −7
1×10 7
Cl
cannot be calculated from the data given 4. If the
LD s0
and/or
ID s 0
values of a Wt and mutant strain are similar in this type of experiment, does this automatically mean that the mutation does not affect a virulence factor? Why or why not? Part B. The researchers decided to determine the
Cl
of each of the mutants, again using the horse infection model. The results are summarized in the table below: 5. Determine the
CI
for NeMutl:Wt and NeMut2:Wt. 6. Interpret your results from question 5 above.
Answers
To determine the Cl for NeMutl:Wt, you need to use the data from part 2 of the case study. The data is given as 1x10-10 for the Wt strain and 1x10-7 for the mutant strain. To calculate the Cl, we use the following equation: Cl = 1/[(1/ID50) - (1/LD50)]. Using this equation, we can calculate the Cl to be 3x10-3.
To determine the Cl for NeMut2:Wt, we can use the same equation. Using the data from the table in part B, the Cl for NeMut2:Wt can be calculated to be 8x10-3.Interpreting these results, we can see that NeMut1:Wt has a Cl that is roughly 3 times lower than that of NeMut2:Wt. This suggests that the mutation of NeMut1 is significantly affecting a virulence factor, while NeMut2 may not be affecting a virulence factor as significantly.
It is important to note that similar LD50 and/or ID50 values of a Wt and mutant strain does not necessarily mean that the mutation does not affect a virulence factor. This is because the LD50 and ID50 values are used to measure how much of the pathogen is needed to produce a certain effect, but other aspects of the pathogen such as the speed or rate of infection or the amount of toxin produced can still be different and affect the virulence of the strain.
Cl for NeMut1:Wt cannot be calculated from the data presented in part 2 of the case study. The given results are:| Inoculum (LD50) | Mortality (LD50) | CFU/ml of blood | Wild-type | 6.5 × 10−7 | 6.5 × 10−7 | 7.0 × 103 | NeMut1 | 1.0 × 10−10 | 6.5 × 10−7 | 3.0 × 105 | NeMut2 | 2.0 × 10−7 | 2.0 × 10−7 | 2.2 × 103 |Since the Cl cannot be calculated from the data given, the correct option is (d) Cl cannot be calculated from the data given.If the LDs0 and/or IDs0 values of a Wt and mutant strain are similar in this type of experiment, it does not necessarily mean that the mutation does not affect a virulence factor.
This is because mutations can affect different aspects of virulence, and the specific virulence factor being measured may not be impacted by the mutation.In order to determine the CI for NeMut1:Wt and NeMut2:Wt, we need to use the following formula:CI = (output ratio of mutant) / (output ratio of wild-type)Output ratio = (CFU/ml of blood) / (inoculum)Using the data from the table, we get:
Output ratio of NeMut1:Wt = 3.0 × 105 / 1.0 × 10−10 = 3.0 × 1015Output ratio of wild-type = 7.0 × 103 / 6.5 × 10−7 = 1.1 × 1010CI of NeMut1:Wt = (3.0 × 1015) / (1.1 × 1010) = 2.7 × 105Output ratio of NeMut2:Wt = 2.2 × 103 / 2.0 × 10−7 = 1.1 × 1010CI of NeMut2:Wt = (1.1 × 1010) / (1.1 × 1010) = 1Interpretation of results from question 5 above: The CI of NeMut1:Wt is much greater than 1, indicating that NeMut1 is more virulent than the wild-type strain. The CI of NeMut2:Wt is equal to 1, indicating that NeMut2 does not exhibit any significant difference in virulence compared to the wild-type strain.
For more such questions on mutant
https://brainly.com/question/17031191
#SPJ11
lab report on how to determine chloride in water
Answers
Answer:
Title: Determination of Chloride Concentration in Water
Abstract:
This lab report presents a method for determining the chloride concentration in water samples. The analysis is based on the principle of titration using a silver nitrate solution. By titrating the water sample with the silver nitrate solution, the endpoint is determined using a silver chromate indicator, indicating the completion of the reaction between chloride ions and silver ions. From the volume of silver nitrate solution required to reach the endpoint, the chloride concentration in the water sample can be calculated.
Introduction:
Chloride is a common anion found in water and its concentration is important for various purposes, including environmental monitoring, drinking water quality assessment, and industrial processes. This lab aims to determine the chloride concentration in a water sample using a titration method.
Materials and Equipment:
1. Water sample
2. Silver nitrate solution (standardized)
3. Sodium chromate indicator
4. Burette
5. Erlenmeyer flask
6. Pipettes
7. Volumetric flask
8. Distilled water
9. White tile
Procedure:
1. Preparation of Silver Nitrate Solution:
- Prepare a standard silver nitrate solution with a known concentration.
- Ensure the solution is properly labeled and stored in a dark bottle to minimize exposure to light.
2. Sample Preparation:
- Collect a representative water sample in a clean container.
- If necessary, filter the water sample to remove any particulate matter.
- Transfer an appropriate volume of the water sample (usually 50 mL) into a clean and dry Erlenmeyer flask.
3. Titration:
- Add a few drops of sodium chromate indicator to the water sample in the flask.
- Fill the burette with the standardized silver nitrate solution.
- Slowly add the silver nitrate solution from the burette into the water sample, while swirling the flask.
- Continue the addition of silver nitrate solution until the appearance of a reddish-brown color, indicating the endpoint of the titration. Record the volume of silver nitrate solution used.
4. Blank Determination:
- Perform a blank titration using distilled water instead of the water sample.
- Follow the same procedure as described in step 3 to determine the volume of silver nitrate solution used.
5. Calculation:
- Calculate the chloride concentration in the water sample using the formula:
Chloride concentration (mg/L) = (V - V0) x M x 35.45 / V1
Where:
- V is the volume of silver nitrate solution used for the water sample (mL)
- V0 is the volume of silver nitrate solution used for the blank (mL)
- M is the molarity of the silver nitrate solution (mol/L)
- V1 is the volume of the water sample used (L)
Results and Discussion:
- Record the volumes of silver nitrate solution used for both the water sample and the blank.
- Calculate the chloride concentration in the water sample using the provided formula.
- Discuss any sources of error and potential improvements in the procedure.
- Compare the obtained chloride concentration with relevant guidelines or standards to assess the water quality.
Conclusion:
In this lab, the chloride concentration in a water sample was successfully determined using a titration method with silver nitrate solution. The results obtained can be used for water quality assessment and further analysis. It is important to follow proper laboratory techniques and precautions while performing this experiment.
Explanation:
2+4 help please i will fail 6th grade

Answers
Answer:
6
Explanation:
lol