a) Use the kinetic molecular theory to explain why volume and pressure are inversely related.
b) One mole of Kr and one mole of N2are placed in separate flasks having the same volume and temperature. Using the KMT,
(i) Compare the pressure of the gases in their containers
(ii) compare the rms velocities
(iii) compare the kinetic energies of the gases
Answers
Volume and pressure are inversely related because according to the kinetic molecular theory, the volume of a gas is directly proportional to the average kinetic energy of its particles, while the pressure of a gas is directly proportional to the frequency and force of collisions between its particles and the container. As the volume decreases, the particles become more crowded and collide with the container more frequently and with more force, resulting in an increase in pressure.
(i) The pressure of the gases in their containers will be the same as they are at the same temperature and have the same volume.
(ii) According to the kinetic molecular theory, the root-mean-square velocity (vrms) of a gas is proportional to the square root of its temperature and inversely proportional to the square root of its molar mass. Since both Kr and N2 are at the same temperature and have the same volume, their vrms values will be the same.
(iii) The kinetic energy (KE) of a gas is directly proportional to the square of its vrms and its molar mass. Since both gases have the same temperature, the vrms of each gas will be the same. However, since the molar mass of Kr is greater than that of N2, Kr will have a higher KE than N2.
To learn more about energy click here, https://brainly.com/question/8630757
#SPJ11
Related Questions
How can you tell if a substance is an acid or base?
Answers
Answer:
To determine whether a substance is an acid or a base, count the hydrogens on each substance before and after the reaction. If the number of hydrogens has decreased that substance is the acid (donates hydrogen ions). If the number of hydrogens has increased that substance is the base (accepts hydrogen ions
On a heating curve a plateau corresponds to A) a change in temperature of a liquid. B) a change in temperature of a solid. C) a change in temperature of a gas. D) a change of state. E) the solid being broken into smaller pieces.
Answers
17 Which of the following options correctly describe the general characteristics and behavior of aromatic hydrocarbons?
They are unsaturated compounds
They tend to undergo substitution reactions
Answers
As a result, they are more susceptible to attack by nucleophiles that can replace one of the substituents on the ring. For example, halogens, nitro groups, and alkyl groups can all be substituted onto the benzene ring of an aromatic hydrocarbon through a substitution reaction.
The general characteristics and behavior of aromatic hydrocarbons are correctly described by the following options:i. They are unsaturated compoundsii. They tend to undergo substitution reactions.Explanation:Aromatic hydrocarbons are organic compounds that contain at least one benzene ring in their structure. They are cyclic, planar, unsaturated, and follow Huckel’s rule. Huckel’s rule states that an aromatic compound must contain a planar, cyclic, and fully conjugated pi-electron system with 4n+2 pi electrons, where n is any integer. These electrons form a delocalized pi-electron cloud above and below the plane of the ring. This cloud is responsible for the aromaticity of the compound, which gives it its stability and unique reactivity.Aromatic hydrocarbons are unsaturated compounds because they contain double bonds between some of their carbon atoms. These double bonds are part of the conjugated pi-electron system that gives the compound its aromatic character. Aromatic hydrocarbons tend to undergo substitution reactions because the pi-electron cloud above and below the plane of the ring makes the carbon atoms more electrophilic. As a result, they are more susceptible to attack by nucleophiles that can replace one of the substituents on the ring. For example, halogens, nitro groups, and alkyl groups can all be substituted onto the benzene ring of an aromatic hydrocarbon through a substitution reaction.
To know more about substitution visit:
https://brainly.com/question/29383142
#SPJ11
(01.02 LC) Which of the following is the unit of length in the metric system?
Group of answer choices
A:Foot
B:Inch
C:Meter
D.Mile
Answers
Answer:
Meter.
Explanation:
A foot is a American standard. Inch is also. A Mile is for a American. Kilometers are the metric. That leaves only the Meter.
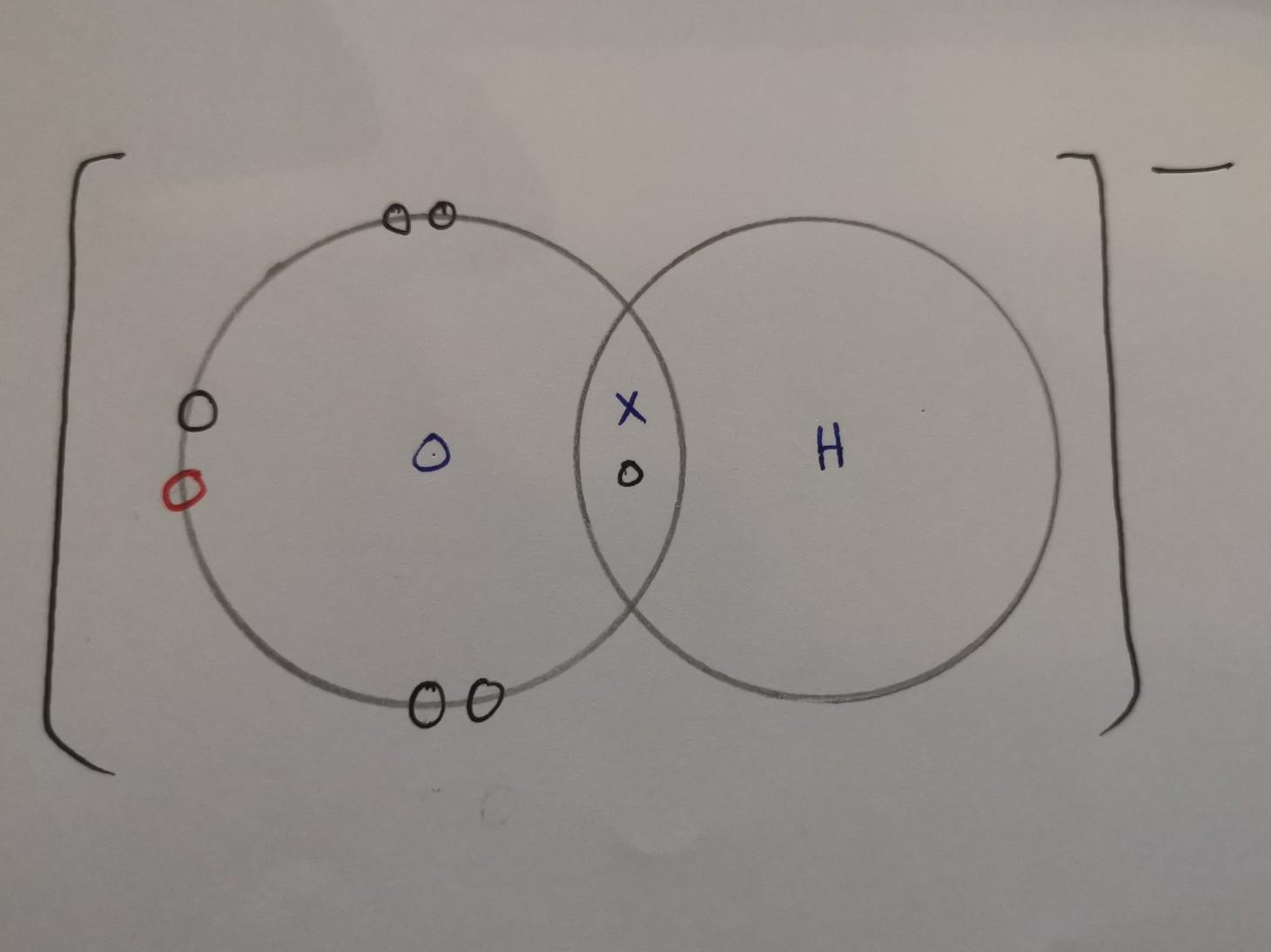
Draw the electron dot structure of the hydroxide ion (OH-).
Answers
Answer:
X = electrons from hydrogen
O (black) = electrons from oxygen
O (red) = electrons from when it was connected to a metal atom, hence why it has a negative charge
Hi, i am almost done with my assignment, 2.11 Unit test chemistry of life, could i get help from someone who knows this type of questions? would get 5 stars and a few extra points well

Answers
A Scientist test a small section of a carrot root to determine the type of molecules were present. the results showed that the molecule contained carbon , hydrogen , and oxygen . the option d is correct (d) both (a) and (c)
(a)the molecule are a type of carbohydrate (c) the carrot contains some type of sugar.
Carrot is one of the important root vegetable rich in bioactive compounds. carbohydrates are made up of carbon , hydrogen and oxygen. carbohydrates are sugar molecules. The main three carbohydrates : sugar , starches and fiber. The carrot vegetable we eat is the taproot of carrot plant. The sucrose is major transport and storage sugar but it convert into starch. Carbohydrates provides energy for living things.
Thus, A Scientist test a small section of a carrot root to determine the type of molecules were present. the results showed that the molecule contained carbon , hydrogen , and oxygen . the option d is correct (d) both (a) and( c)
(a)the molecule are a type of carbohydrate
(c) the carrot contains some type of sugar.
To learn more about carbohydrate here
https://brainly.com/question/14614055
#SPJ1
¿Cuántos moles de sal hay en
13. 8
g
13. 8g13, point, 8, start text, g, end text de cloruro de sodio?
Answers
There are 0.235 moles of salt in 13.8 g of chloride of sodium.
Moles and grams are related by the molecular weight of a compound.
The molecular weight of a substance is the sum of the atomic weights of all the atoms present in its chemical formula. For chloride of sodium, NaCl, the atomic weight of Na is 23.0 g/mol, and the atomic weight of Cl is 35.5 g/mol.
The molecular weight of NaCl is, therefore, 58.5 g/mol.
To calculate the number of moles in 13.8 g of NaCl, we need to divide the mass of the substance by its molecular weight. 13.8 g / 58.5 g/mol = 0.235 moles of NaCl.
For more such questions on moles
https://brainly.com/question/29367909
#SPJ8
1. Iron metal reacts with oxygen to give iron (III) oxide according to the following reaction.
4Fe +30₂
2Fe₂O,
-
a. An ordinary iron nail (assumed to be pure iron) that contains 2.8 g of iron (MM-56
g/mol) reacts in an environment where there is 1.28 g oxygen (MM-32 g/mol). Show a
calculation to determine the limiting reactant in this reaction. (3 pts)
b. How many grams of Fe,O, (MM-160 g/mol) will be formed in the reaction? (3 pts)
c. How many grams of the excess reactant remains after the reaction stops? (3 pts)
Answers
a. Oxygen is the limiting reactant.b. 4.32 g Fe2O3 are formed.c. 0.96 g of excess oxygen remains after the reaction stops.
We are given the balanced chemical equation for the reaction between iron and oxygen. The balanced chemical equation is:4Fe + 3O2 → 2Fe2O3Now, we need to calculate the number of moles of each reactant that we have. The molar mass of iron (Fe) is 56 g/mol, and the molar mass of oxygen (O2) is 32 g/mol.Number of moles of iron:2.8 g ÷ 56 g/mol = 0.05 molNumber of moles of oxygen:1.28 g ÷ 32 g/mol = 0.04 molAccording to the balanced equation, 4 moles of iron react with 3 moles of oxygen. So, the mole ratio of iron to oxygen is 4:3. To determine which reactant is limiting, we need to compare the actual mole ratio to the required mole ratio.Actual mole ratio:0.05 mol iron ÷ 0.04 mol oxygen = 1.25:1Required mole ratio:4 mol iron ÷ 3 mol oxygen = 1.33:1Since the actual mole ratio is less than the required mole ratio, we can conclude that oxygen is the limiting reactant.Now that we have identified the limiting reactant, we can use the balanced equation to determine the amount of product formed.According to the balanced equation, 4 moles of iron react to form 2 moles of Fe2O3. The molar mass of Fe2O3 is 160 g/mol.Moles of Fe2O3 formed:0.04 mol oxygen × (2 mol Fe2O3 ÷ 3 mol oxygen) = 0.027 molFe2O3Mass of Fe2O3 formed:0.027 mol × 160 g/mol = 4.32 g Fe2O3 We know that oxygen is the limiting reactant, which means that there is some iron left over after the reaction stops. To determine how much iron is left over, we first need to determine how much iron reacted. Since 4 moles of iron react with 3 moles of oxygen, the number of moles of iron that reacted is:Moles of iron reacted:0.04 mol oxygen × (4 mol iron ÷ 3 mol oxygen) = 0.053 mol ironMass of iron reacted:0.053 mol iron × 56 g/mol = 2.98 g ironTo determine how much excess oxygen is left over, we can subtract the amount of oxygen that reacted from the total amount of oxygen we started with:Mass of excess oxygen:1.28 g - (0.04 mol oxygen × 32 g/mol) = 0.96 g excess oxygenAnswer:a. Oxygen is the limiting reactant.b. 4.32 g Fe2O3 are formed.c. 0.96 g of excess oxygen remains after the reaction stops.For such more questions on Oxygen
https://brainly.com/question/2111051
#SPJ8
Uranium-235, uranium-238 and uranium-239 are different
A) elements.
B) ions of the same element.
C) isotopes of the same element.
D) none of the above
Answers
Answer:
C
Explanation:
they are of same element but different mass no.
in chemistry language we call them isotopes
Please answer the following question using the data below: H2O vapor content: 13 grams H2O vapor capacity: 52 grams at 25 degrees Celsius 13 grams at 10 ∘
C 52 grams at 30 ∘
C What is the dew point for the conditions listed above? LCL 3π5 25C Relative Humidity =100%
Answers
Given data:H2O vapor content: 13 gramsH2O vapor capacity: 52 grams at 25 degrees Celsius 13 grams at 10∘C52 grams at 30∘CFormula used to find the dew point:$$\dfrac{13}{52}=\dfrac{(A*3\pi)/(ln100)}{(17.27-A)}$$$$\frac{1}{4}=\dfrac{(A*3\pi)/(ln100)}{(17.27-A)}$$
Where A is the constantDew Point:It is the temperature at which air becomes saturated with water vapor when the temperature drops to a point where dew, frost or ice forms. To solve this question, substitute the given data into the formula.$$13/52=\dfrac{(A*3\pi)/(ln100)}{(17.27-A)}$$$$13(17.27-A)=3\pi A(ln100)$$By simplifying the above expression, we get$$A^2-17.27A+64.78=0$$Using the quadratic formula, we get$$A=9.9,7.4$$
The dew point is 7.4 since it is less than 10°C.More than 100:The term "More than 100" has not been used in the question provided.
To know more about temperature visit:
https://brainly.com/question/7510619
#SPJ11
You're paid $25 per hour for your job. How much would you earn in cents per second?
Answers
Answer:
0.694 cents per second
Explanation:
25x100=2500 cents per hour, 2500/60 = 41.67 per minute and 41.67/60=0.694 cents per second
HELPPPP!!!! SCIENCE!!!!
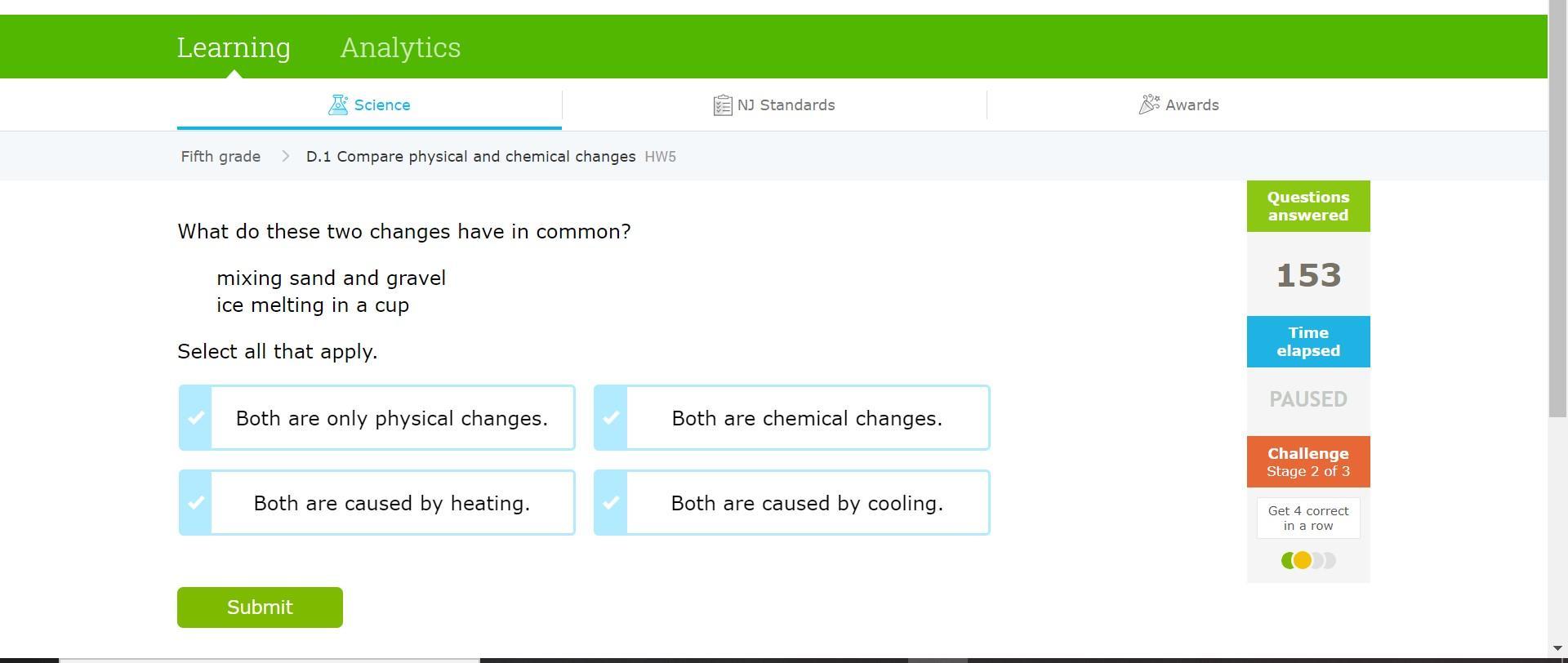
Answers
Answer:
Both are only physical changes
In an acid-base reaction involving neutral base B, what will be the conjugate acid Select the correct answer below , a. h20 b. H30 c. Oh- d hb+
Answers
In an acid-base reaction involving a neutral base B, the conjugate acid will be H₃O⁺. Option B is correct.
In an acid-base reaction involving a neutral base B, the conjugate acid will be formed by the addition of a proton (H⁺) to the base.
H₂O; Water (H₂O) is not a base but can act as an acid in certain reactions. It can donate a proton to form the hydroxide ion (OH⁻), making it a conjugate base rather than a conjugate acid.
H₃O⁺; The hydronium ion (H₃O⁺) is formed when a proton (H⁺) is added to water (H₂O). It is commonly found in aqueous acidic solutions and can act as an acid by donating a proton. Therefore, H₃O⁺ is the correct answer as it represents the conjugate acid in this acid-base reaction.
OH⁻; The hydroxide ion (OH⁻) is a base, not an acid. It accepts a proton (H⁺) to form water (H₂O) in basic solutions. OH⁻ would be the conjugate base, not the conjugate acid.
Hence, B. is the correct option.
To know more about conjugate acid here
https://brainly.com/question/8908134
#SPJ4
--The given question is incorrect, the correct question is
"In an acid-base reaction involving neutral base B, what will be the conjugate acid Select the correct answer below , A) H₂O B). H₃0 C). OH⁻."--
What two of the following organisms are secondary consumers in this food web?
Answers
Secondary consumers are organisms that primarily feed on herbivores or other primary consumers.
They occupy the next trophic level above the primary consumers in a food web. They obtain energy by consuming the primary consumers and play an important role in regulating the population of herbivores.
Examples of commonly observed secondary consumers include:
Carnivorous mammals: Animals such as wolves, lions, and tigers that feed on herbivores like deer, zebras, or gazelles.
Birds of prey: Species like eagles, hawks, and owls that consume small mammals, reptiles, or other birds.
Carnivorous fish: Fish like pike, barracuda, or bass that prey on smaller fish or aquatic invertebrates.
Predatory insects: Insects such as spiders, mantises, or dragonflies that feed on other insects, including herbivorous insects.
In a specific food web, the identification of secondary consumers would depend on the specific organisms present and their feeding interactions. It would be necessary to analyze the trophic relationships among the organisms in the food web to determine the secondary consumers accurately.
For more such questions on Secondary consumers visit:
https://brainly.com/question/28631974
#SPJ8
what are the factors affecting gravity?
Answers
Gravity, as a fundamental force of nature, is influenced by several factors. The following are some of the key factors affecting gravity:
Mass: The most significant factor affecting gravity is the mass of the objects involved. According to Newton's law of universal gravitation, the gravitational force between two objects is directly proportional to the product of their masses. Greater mass leads to a stronger gravitational force.Distance: The distance between two objects also plays a crucial role in the strength of gravity. According to the inverse square law, the gravitational force decreases as the distance between objects increases. As objects move farther apart, the gravitational attraction between them weakens.Gravitational Constant: The gravitational constant, denoted by G, is a fundamental constant in physics that determines the strength of the gravitational force. It is a universal constant and does not change, affecting the overall magnitude of gravity.Shape and Distribution of Mass: The distribution of mass within an object can influence the gravitational field it generates. Objects with a more compact and concentrated mass distribution will have a stronger gravitational pull compared to those with a more spread-out mass distribution.External Influences: Gravity can be influenced by external factors such as nearby celestial bodies or the presence of other forces. For example, the gravitational interaction between the Earth and the Moon affects tides on Earth's surface.how much energy is needed to convert 120g of ice at -35°C to steam at 150°C?
Answers
During a reaction, the enthalpy of formation of an intermediate is 90.3 kJ/mol. During the reaction, 2 moles of the intermediate are formed as a reactant. What is the enthalpy value for this step of the reaction?
A. -180.6 kJ
B. 90.3 kJ
C. -90.3 kJ
D. 180.6 kJ
Answers
Answer:
180.6 kJ
Explanation:
The enthalpy of reaction refers to the energy absorbed or released during a reaction. If heat is absorbed in a reaction, the enthalpy of reaction is positive. If heat is released in a reaction, the enthalpy of reaction his negative.
Since the energy absorbed when one mole of the intermediate is formed is 90.3 kJ/mol, then, when two moles of intermediate is formed, 2 × 90.3 kJ/mol = 180.6 kJ of energy is absorbed.
why is time an independent variable
Answers
If 0.08 moles of Fe are
reacted, how many moles of
H, are formed?
Answers
Based on the balanced equation, if we have 0.08 moles of Iron (Fe), we can conclude that 0.08 moles of Hydrogen (H₂) will be formed as well.
How to Calculate Mole in a Chemical EquationFirst we need to balance the chemical equation for the reaction involving Iron (Fe) and Hydrogen (H).
The reaction will be between Fe and HCl (hydrochloric acid) to produce hydrogen gas (H₂) and iron chloride (FeCl₂):
Fe + 2HCl → FeCl₂ + H₂
From the balanced equation, we can see that for every 1 mole of Fe reacted, 1 mole of H₂ is formed. Therefore, if we have 0.08 moles of Fe, we can conclude that 0.08 moles of H₂ will be formed as well.
Learn more about moles here:
https://brainly.com/question/15356425
#SPJ1
Imagine you fill a balloon with air to a volume of 240 atm. Initially the air temperature i 25* C and the air preure i 1. 0 atm. You carry the balloon with you up a mountain where the air preure i 0. 75 atm and the temperature i 25* C. Solve for the new volume of the balloon
Answers
The new volume of the balloon is V2 = 180 atm. The ideal gas law can be used to calculate the volume of a gas if the pressure, temperature, and number of moles are known.
This equation is PV=nRT, where P is the pressure, V is the volume, n is the number of moles, R is the universal gas constant, and T is the temperature. In this case, the initial pressure is 1.0 atm, the temperature is 25° C, and the number of moles is unknown. However, the initial volume is known, so the equation can be rearranged to solve for n:
V1P1=nRT
n=V1P1/RT
n=(240 atm1.0 atm)/(8.314 J/(molK)*298 K)
n=7.7 moles
The new volume of the balloon can then be calculated using the ideal gas law:
P1V1=P2V2
1.0 atm * 240 atm = 0.75 atm * V2
V2 = 180 atm
Learn more about volume of the balloon:
https://brainly.com/question/28601285
#SPJ4
Explain what a scientist is
Answers
but the role of a scientist is..: scientists use their resources to bring out change, wether it is change in medicine, improvements in the environment, or creating efficient fuels
I rlly hope this helps tysm
How much of a 200 g sample of Gold is left after 13.5 days if its half-life is 2.70 days?
Answers
Answer: 3.2
Explanation:
Does Fe(NO3)2 react with Pb
Answers
Answer:you only need one to balance Fe(NO3)2 react with Pb
Explanation:
(Please help I will give brainliest to whoever answers)Classify each of the following as either an element, compound, mechanical mixture, solution, colloid or suspension
a. Gold
b. Kool-aid
c. Carbon dioxide
d. Tomato juice
e. Milk
f. Salad
Answers
Answer:
element:a,c
compound:f
mechanical mixture
solution:b
colloid:e
suspension:d
Explanation:
What is combustion??
Answers
Answer:
Measure of how much heat is given off when a substance is burned
Explanation:
hope this helps
The reaction of an acid and a base produce water and a
Answers
Answer:
The answer is " salt".
Explanation:
Hey there!
When any acids and base react with eachother, they forms water and respective salts.
For example:
HCl + NAOH = NACl + H2O.
Hope it helps!
convert the following moles to the number of representative particles
0.250 mol silver to silver atoms
Answers
There are 1.51 * 10^23 particles
How do you get the number of particles from number of moles?To get the number of particles from the number of moles, you can use Avogadro's number, which is the number of particles in one mole of a substance. Avogadro's number is approximately 6.022 x 10^23 particles per mole.
To convert the number of moles to the number of particles, you can multiply the number of moles by Avogadro's number. Mathematically, this can be expressed as:
number of particles = number of moles x Avogadro's number
Given that;
1 mole would contain 6.02 * 10^23 particles
0.25 mol = 0.25 * 6.02 * 10^23 particles/ 1 mole
= 1.51 * 10^23 particles
Learn more about moles:https://brainly.com/question/26416088
#SPJ1
(T/F) reactive oxygen intermediates and reactive nitrogen intermediates produced by phagocytic cells are effective in killing invading microorganisms
Answers
The statement 'reactive oxygen intermediates and reactive nitrogen intermediates produced by phagocytic cells are effective in killing invading microorganisms' is true.
Phagocytic cells are the kind of cells that consume or destroy microorganisms by producing substances that are harmful to them, according to the study of biology.
The phagocytic cells produce oxygen and nitrogen intermediates, and these intermediates are reactive enough to bind to the chemicals in the microbe and inhibit their function. The microorganism dies when its activities are impeded to the point where it is unable to carry out the fundamental functions of life. Thus, the above statement is accurate.
To learn more about phagocytic cells, visit the link below:
brainly.com/question/29832465
#SPJ4
How is Hess's law used to calculate the enthalpy of a reaction?
A. The enthalpy is obtained from the difference in final and initial reactions in a path.
B. Enthalpies from similar equations are substituted for unknown reaction enthalpies. C. The final equation in a reaction path provides the enthalpy for the desired reaction.
D. The desired enthalpy is obtained through adding intermediate reactions together.
Answers
Considering the Hess's Law, the desired enthalpy is obtained through adding intermediate reactions together. (option D)
Hess's LawHess's law, also called Law can also be called Law of additivity of the enthalpies of reaction, it is a useful method when it is not possible to calculate the enthalpies of reaction from the enthalpies of formation, or in reactions in which the reaction enthalpy cannot be determined experimentally because it is very slow or very explosive.
Hess's law states that when reactants are converted to products, the enthalpy change is the same whether the reaction takes place in one step or in a series of steps.
In this way, Hess's Law determines that when a chemical reaction can be expressed as an algebraic sum of other reactions, its reaction enthalpy is equal to the sum of the enthalpies of the partial reactions. In this way, it allows thermochemical equations to be treated as algebraic equations, and can be added, subtracted, or multiplied by a number to find the desired thermochemical equation.
SummaryHess's Law states that the sum of the ∆H of each stage of the reaction will give us a value equal to the ∆H of the reaction when it occurs in a single stage.
That is, the desired enthalpy is obtained through adding intermediate reactions together. (option D)
Learn more about Hess'Law:
brainly.com/question/5976752
brainly.com/question/13707449
brainly.com/question/13707449
brainly.com/question/6263007
brainly.com/question/14641878
brainly.com/question/2912965
#SPJ1
Light of frequency 9.62 x 1014 Hz
(312 nm) strikes a surface of calcium
(Wo = 2.90 eV). How much kinetic
energy does the liberated electron
have in Joules?
Answers
The kinetic energy of the liberated electron is 1.73 x 10⁻¹⁹ J.
What is the energy of the light?
The total energy of the light is the sum of the kinetic energy of the electron and work function of the metal.
E = K.E + Ф
where;
K.E is the kinetic energy of the liberated electronФ is the work function of the metalThe total energy of the photons of light is calculated as follows;
E = hf
where;
f is the frequency of the lighth is Planck's constantE = (6.626 x 10⁻³⁴) x (9.62 x 10¹⁴)
E = 6.37 x 10⁻¹⁹ J
1 eV = 1.6 x 10⁻¹⁹ J
E = 6.37 x 10⁻¹⁹ J = 3.98 eV
The kinetic energy of the liberated electron is calculated as;
E = K.E + Ф
K.E = E - Ф
K.E = 3.98 eV - 2.9 eV
K.E = 1.08 eV = 1.73 x 10⁻¹⁹ J
Learn more about energy of photon here: https://brainly.com/question/27856390
#SPJ1